
Concept explainers
Interpretation: The systematic name of glycerol should be determined.
Concept Introduction: The functional group that alcohol contains is hydroxyl (-OH) group. There are three types of alcohols that is primary, secondary, and tertiary alcohol classified on the basis of number of carbons to which the hydroxyl group is attached.
Answer to Problem 65A
The systematic name of glycerol is propane-1,2,3-triol.
Explanation of Solution
In order to give the IUPAC name to the alcohol following steps are followed:
1. The parent (longest)
2. The ending of the parent chain from alkane (-e) is changed to -ol and the number is used to locate the -OH group of alcohol.
3. Name should be written in alphabetical order and numbering should be done in such a way that hydroxyl group gets lowest number.
The given structure is:
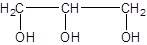
The parent chain in the given structure is of 3 carbon atoms, propane. Numbering is done in such a way that hydroxyl (-OH) group gets lower number as:
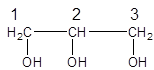
So, the systematic name will be: propane-1,2,3-triol.
Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





