
Concept explainers
Interpretation:
The Kiliani–Fischer reaction for the synthesis of epimeric aldoses is to be outlined and Fischer projection of
Concept introduction:
舧 A carbohydrate is a
舧
舧 Carbohydrates are oxidized by
舧 Aldaric acids are carbohydrates having two carboxylic acids. They are formed due to oxidation reaction of aldoses with dilute
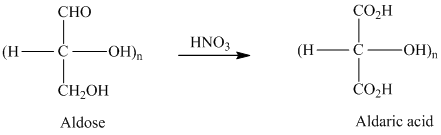
舧 The molecules that are nonsuperimposable or not identical with their mirror images are known as chiral molecules.
舧 A pair of two mirror images that are nonidentical is known as enantiomers, which are optically active.
舧 The stereoisomers that are nonsuperimposable on each other and not mirror images of each other are known as diastereomers.
舧 The achiral compounds in which plane of symmetry is present internally and consists of chiral centres are known as meso compounds, but they are optically inactive.
舧 Compounds that have a plane of symmetry tend to exist in meso forms. A meso form arises when the two stereoisomers produce superimposable images, and hence, compounds having meso forms are optically inactive.
舧 The Kiliani-Fischer reaction is used for increasing the number of carbon atoms of the respective aldoses, thereby, lengthening the carbon chain of the compound. It is an effective and economic process for synthesizing monosaccharides through the hydrolysis of a cyanohydrin. Thus, it lengthens the carbon chain of the aldose by one carbon atom. It is used to produce epimers of higher aldoses from a lower aldose, that is, a single aldose produces a mixture of two diastereomeric sugars called epimers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Organic Chemistry
- Treatment of cholesterol with mCPBA results in formation of a single epoxide A, with the stereochemistry drawn. Why isn’t the isomeric epoxide B formed to any extent?arrow_forwardAssume that for your Biochemistry practical, you were asked to synthesise D-Galactose. You went to chemical storage room to take some D-lyxose to use as the starting material. But there you found that labels had fallen off from the bottles containing D-lyxose and D-xylose. How could you determine which bottle contains D- lyxose?arrow_forwardPredict the reactions ofcarbohydrates in acidic and basicsolutions, and with oxidizingand reducing agentsarrow_forward
- An aldose A, is reduced by sodium borohydride to an optically inactive alditol B. The Ruff degredation of A forms C. Oxidation of C by nitric acid generates the optically inactive diacid D. The ruff degreation of C forms D-glyceraldehyde. draw the structures for compounds A through D and discuss a mechanism for the reduction step using sodium borohydridearrow_forwardWhen the gum of the shrub Sterculia setigera is subjected to acidic hydrolysis, one of the water-soluble components of thehydrolysate is found to be tagatose. The following information is known about tagatose:(1) Molecular formula C6H12O6(2) Undergoes mutarotation.(3) Does not react with bromine water.(4) Reduces Tollens reagent to give d-galactonic acid and d-talonic acid.(5) Methylation of tagatose (using excess CH3 I and Ag2O) followed by acidic hydrolysis gives1,3,4,5-tetra-O-methyltagatose.(a) Draw a Fischer projection structure for the open-chain form of tagatose.(b) Draw the most stable conformation of the most stable cyclic hemiacetal form of tagatosearrow_forwardBradykinin is a linear nonapeptide released by blood plasma globulin in response to a wasp sting. It is a very potent pain-causing agent. Its constituent amino acids are 2R, G, 2F, 3P, S. The sue of 2,4-dinitrofluorobenzene and carboxypeptidase shows that both terminal residues are arginine. Partial hydrolysis of bradykinin gives the following di- and tripeptides: FS + PGF +PP + SPF + FR + RParrow_forward
- Thalidomide is a chiral molecule and it was identified that the R-isomerproduced the sedative properties whereas the S-isomer produced the teratogenic effects.Identify the chiral centre in the thalidomide molecule and, using your knowledge of enolisation, illustrate mechanistically, and explain, why there would be no benefit to a patient taking the chirally pure R-isomer of the drug to avoid the sideeffects of the other enantiomer.arrow_forwardIs fexofenadine chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardThoroughly explain why (a)malthose is a reducing sugar while trehalose is not based on their structures. (b)Why is trehalose very resistant to acid hydrolysis while maltose can be acid-hydrolyzed with ease. Give clear explanations.arrow_forward
- Emil Fischer synthesized l-gulose, an unusual aldohexose that reduces to give d-glucitol.Suggest a structure for this l sugar, and show how l-gulose gives the same alditol asd-glucose. (Hint: d-Glucitol has ¬CH2OH groups at both ends. Either of these primaryalcohol groups might have come from reduction of an aldehyde.)arrow_forwardIllustrate the treatment of methyl α-D-glucopyranoside with aqueous acid forms a mixture of α- and β-D-glucose and methanol ?arrow_forwardSuggest how you would separate the free l-amino acid from its acylated d enantiomerarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

