
Concept explainers
(a)
Interpretation: Whether the indicated cyclohexane derivative is chiral or not should be identified.
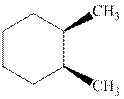
Concept introduction: Four kinds of symmetry elements that may be present are tabulated as follows:
The enantiomers are identical chemical compounds that have a mirror-image relationship to each other while diastereomers do not hold mirror image relationships. The former are chiral and optically active while the latter can be chiral or achiral.
Any organic compound must have no plane of symmetry in order to be optically active. The compounds with any plane of symmetry are achiral and optically inactive.
(b)
Interpretation: Whether the indicated cyclohexane derivative is chiral or not should be identified.
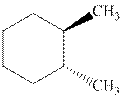
Concept introduction: Four kinds of symmetry elements that may be present are tabulated as follows:
The enantiomers are identical chemical compounds that have a mirror-image relationship to each other while diastereomers do not hold mirror image relationships. The former are chiral and optically active while the latter can be chiral or achiral.
Any organic compound must have no plane of symmetry in order to be optically active. The compounds with any plane of symmetry are achiral and optically inactive.
(c)
Interpretation: Whether the indicated cyclohexane derivative is chiral or not should be identified.
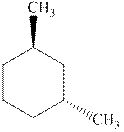
Concept introduction: Four kinds of symmetry elements that may be present are tabulated as follows:
The enantiomers are identical chemical compounds that have a mirror-image relationship to each other while diastereomers do not hold mirror image relationships. The former are chiral and optically active while the latter can be chiral or achiral.
Any organic compound must have no plane of symmetry in order to be optically active. The compounds with any plane of symmetry are achiral and optically inactive.
(d)
Interpretation: Whether the indicated cyclohexane derivative is chiral or not should be identified.

Concept introduction: Four kinds of symmetry elements that may be present are tabulated as follows:
The enantiomers are identical chemical compounds that have a mirror-image relationship to each other while diastereomers do not hold mirror image relationships. The former are chiral and optically active while the latter can be chiral or achiral.
Any organic compound must have no plane of symmetry in order to be optically active. The compounds with any plane of symmetry are achiral and optically inactive.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: Structure and Function
- Consider 1-bromo-2-methylpropane and draw the following. (a) The staggered conformation(s) of lowest energy (b) The staggered conformation(s) of highest energyarrow_forwardKetones react with alcohols to yield products called acetals. Why does the all-cis isomer of 4-tert-butyl-1,3-cyclohexanediol react readily with acetone and an acid catalyst to form an acetal, but other stereoisomers do not react? In formulating your answer, draw the more stable chair conformations of all four stereoisomers and the product acetal for each one.arrow_forwardCyclohexane derivatives exist primarily in the most stable of the available chair conformations. Give the position, axial or equatorial, of each of the three groups shown when the ring is in the most stable chair conformation. If a group divides its time equally between axial and equatorial positions, indicate this with ax/eq.arrow_forward
- When the given alkene undergoes hydroboration followed by oxidation, which would be produced in regards to the products stereochemistry a. A mix of diastereomers b. A single enantiomer c. A racemic mixture d. An achiral productarrow_forwardCalculate the difference in energy between the two conformation of trans_4_tert_butyl_1_methylcyclohexanearrow_forwardWhat is the degree of unsaturation for compoundC11H7ClO?arrow_forward
- How many gauche interactions are present in the most stable chair conformation of 1,1,2-trimethylcyclohexane?arrow_forwardHow many unique stereoisomers exist for 1,2,3,4,5-pentachlorocyclohexane?arrow_forwardUsing what you know about the conformational energetics of substituted cyclohexanes, predict which of the two decalin isomers is more stable. Explain your reasoning.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


