
Concept explainers
Interpretation:Stereocenters in the indicated chiral molecules should be marked and any one of the stereoisomers should be drawn along with absolute stereochemistry as R or S.
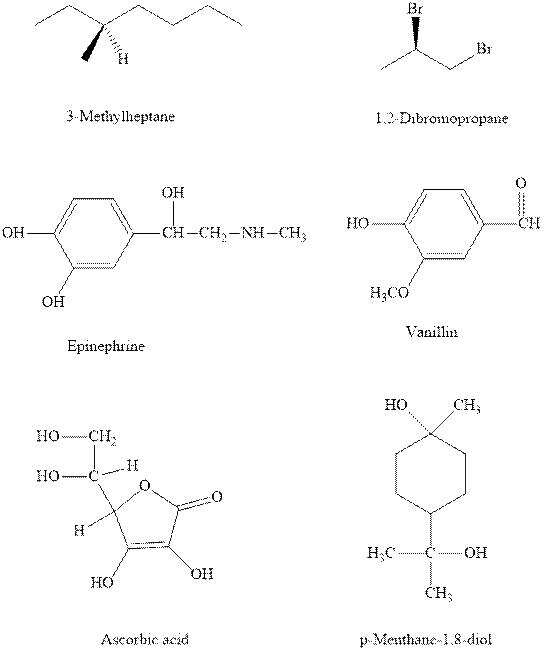
Concept introduction:Chiral carbon is any stereocenter attached to four different alkyl substituents. It is also known as stereocenter. If any two of the substituent happen to be similar the center is regarded as achiral. However, the most essential criteria that help to distinguish a chiral or non-chiral system is presence of any symmetry element. If any plane center or axis of symmetry is identified it makes molecule achiral and optically inactive.
In order to assign absolute configuration of R and S, Cahn − Ingold − Prelog rules are used and the first step is to assign the priority order on the basis of
If the groups read from highest towards least in clockwise fashion then R is assigned to the stereocenter if the rotation is anticlockwise then S is assigned at the configuration.
The Fischer projection is written along with the priorities assigned and groups are interchanged between adjacent places so as to obtain lowest priority group at the bottom or lowest priority.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: Structure and Function
- Considerthe image below. a. Draw the mirror image of structure I and label it as structure II.b. Are I and II chiral?c. What kind of stereocenters do they represent?arrow_forwardIdentify the absolute configuration of the centers of chirality in each of the following compounds as R or S. If two centers of chirality are present, indicate the configurations as: RR, SS, RS, or SR.arrow_forwardLabel each of the following chiral centers as R or S.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

