
Concept explainers
(a)
Interpretation: The type of structures needs to be determined.

Concept Introduction:Structural isomers have the same chemical formula, but the atoms arrangements are different in both structures.
(b)
Interpretation: The type of structures needs to be determined.
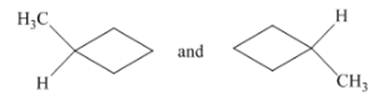
Concept Introduction:The identical structures have same groups, but different positions.
(c)
Interpretation: The type of structures needs to be determined.
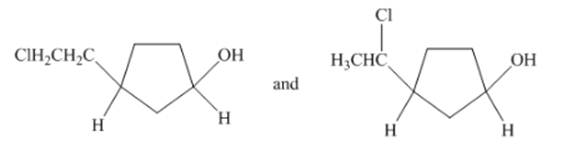
Concept Introduction:Structural isomers have same molecular formula, but the arrangements of atoms and groups are different.
(d)
Interpretation: The type of structures needs to be determined.
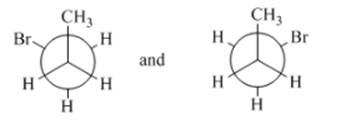
Concept Introduction:Conformers have the same formula, but the isomers can be interchanged by rotation at an angle.
(e)
Interpretation: The type of structures needs to be determined.
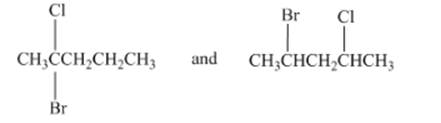
Concept Introduction:Structural isomers have same formula but different arrangements of molecules and atoms.
(f)
Interpretation: The type of structures needs to be determined.

Concept Introduction:Stereoisomer has same molecular formula, but the arrangements of the groups are different in 3-D space.
(f)
Interpretation: The type of structures needs to be determined.

Concept Introduction:Stereoisomer has same molecular formula, but the arrangements of the groups are different in 3-D space.
(g)
Interpretation: The type of structures needs to be determined.
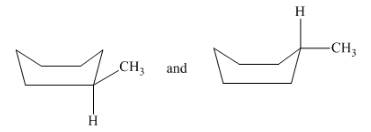
Concept Introduction:Conformers have the same formula, but the groups are arranged in a different manner.
(h)
Interpretation: The type of structures needs to be determined.
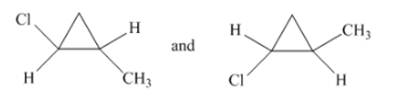
Concept Introduction:Stereoisomer has same molecular formula, but the position of the groups is different in 3-D space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: Structure and Function
- Consider 1-bromo-2-methylpropane and draw the following. (a) The staggered conformation(s) of lowest energy (b) The staggered conformation(s) of highest energyarrow_forwardWould someone be able to explain conformationally locked enantiomers? What types of structures go under this category and are they still optically active?arrow_forwardHow do you account for the difference in energies between the two staggered conformations of 1,2-dichloroethane? How about for the two eclipsed conformations? Draw all four conformations and, on your drawing, indicate sources of strain – torsional, steric (gauche), steric eclipsed.arrow_forward
- Classify attached conformation as staggered or eclipsed around theindicated bond, and rank the conformations in order of increasingstability.arrow_forwardWhich staggered conformation(s) has (have) the lowest energy? Which has (have) the highest energy?arrow_forwardWhat are the differences between stereoisomers, constituional isomers, conformational isomers, and configuration isomers. Please show examplesarrow_forward
- Explain briefly and clearly the following concepts, taking as reference the molecule of n-butane and the corresponding drawings or illustrations. See pages 149-152 of the book Organic Chemistry, sixth edition (J. G. Smith). 4. What is steric hindrance in a conformation? Then draw a picture to illustrate the concept? 5. What is the torsional stress of a conformation? Then draw a picture to illustrate the concept? 6. Describe 1,3-diaxial interaction and illustrate with a specific example.arrow_forwardDefine conformationarrow_forwardHow to identify constitutional isomers from stereoisomers? Can you provide an example? With molecular formula if it's possible?arrow_forward
- Which of the following conformers has the highest energy (the least stable)?Justify your choicearrow_forwardOn the more stable conformations (number one part A drawing), can you label each of the carbons as primary, secondary, tertiary, or quaternary.arrow_forwardCategorize each pair of structures using all the terms that apply: Identical, isomers, constitutional isomers, stereoisomers, enantiomers, and diastereomers.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

