
Concept explainers
a)
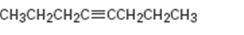
Interpretation:
The product expected during hydration of 4-octyne is to be given.
Concept introduction:
Addition of water to triple bonds occurs following Markovnikov regiochemistry to yield an enol as the product. The enol tautomerizes to give a ketone as the final product. Terminal
To give:
The product expected during hydration of 4-octyne.
b)
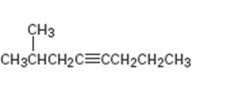
Interpretation:
The product expected during hydration of 2-methyl-4-octyne is to be given.
Concept introduction:
Addition of water to triple bonds occurs following Markovnikov regiochemistry to yield an enol as the product. The enol tautomerizes to give a ketone as the final product. Terminal alkenes and symmetrically substituted internal alkynes yield only one product. Unsymmetrically substituted internal alkynes yield a mixture of ketones as product.
To give:
The products expected during hydration of 2-methyl-4-octyne.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- Acid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardHow would you prepare the following alkyl halides, from alkenes? Write the equation for each reaction indicating substrates, reagents, and conditions.arrow_forwardWhat product do you expect to obtain from each of the following reactions?arrow_forward
- Show how you would synthesize the following compounds from alkyl halides, vinylhalides, and aryl halides containing no more than six carbon atoms.(a) octanearrow_forwardHow many monohalogenation products can be obtained from the following compounds?arrow_forwardProvide a structure for the major products of the reactions and include the steps for each reaction.arrow_forward
- What compound is the expected product upon Markovnikov hydrohalogenation with HBr of the alkene shown below?arrow_forwardWhat is the product of the following reaction? (please provide detailed steps on how you got to the answer) thank you :)arrow_forwardStarting from any aromatic hydrocarbon of your choice, how would you synthesize the following substances? Ortho and para isomers can be separated if necessary.arrow_forward
- What products are obtained by hydration of the symmetrical alkyne shown below? Show the mechanism.arrow_forwardComplete the reaction schemes below providing the products of the reaction schemes below.arrow_forwardPredict the products of the reaction with acetaldehyde with the following reagents?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

