
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.SE, Problem 34AP
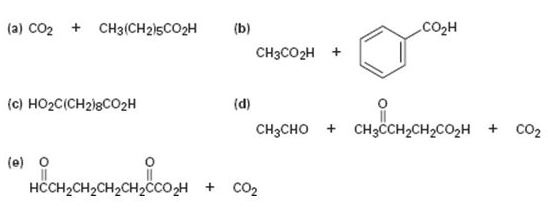
Propose structures for hydrocarbons that give the following products on oxidative cleavage by KMnO4 or O3:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Propose the structures for hydrocarbons (and give the organic reaction) that give the following products on oxidative cleavage with KMnO 4 or O 3 . EXPLAIN
Propose the structures for hydrocarbons (and give the organic reaction) that give the following products on oxidative cleavage with KMnO 4 or O 3 .
Give the expected products of lithium aluminum hydride reduction of the followingcompounds (followed by hydrolysis).(a) butyronitrile (b) N-cyclohexylacetamide
Chapter 9 Solutions
Organic Chemistry
Ch. 9.1 - Prob. 1PCh. 9.1 - Prob. 2PCh. 9.3 - What products would you expect from the following...Ch. 9.4 - Prob. 4PCh. 9.4 - Prob. 5PCh. 9.4 - Prob. 6PCh. 9.4 - Prob. 7PCh. 9.5 - Using any alkyne needed, how would you prepare the...Ch. 9.7 - The pKa of acetone, CH3COCH3, is 19.3. Which of...Ch. 9.8 - Prob. 10P
Ch. 9.8 - Prob. 11PCh. 9.9 - Show the terminal alkyne and alkyl halide from...Ch. 9.9 - Beginning with acetylene and any alkyl halide...Ch. 9.SE - Name the following alkynes, and predict the...Ch. 9.SE - From what alkyne might each of the following...Ch. 9.SE - Prob. 16VCCh. 9.SE - The following cycloalkyne is too unstable to...Ch. 9.SE - Prob. 18MPCh. 9.SE - Assuming that strong acids add to alkynes in the...Ch. 9.SE - Prob. 20MPCh. 9.SE - Prob. 21MPCh. 9.SE - Prob. 22MPCh. 9.SE - Prob. 23MPCh. 9.SE - Prob. 24MPCh. 9.SE - Reaction of acetone with D3O+ yields...Ch. 9.SE - Give IUPAC names for the following compounds:Ch. 9.SE - Draw structures corresponding to the following...Ch. 9.SE - Prob. 28APCh. 9.SE - Prob. 29APCh. 9.SE - Prob. 30APCh. 9.SE - Predict the products from reaction of l-hexyne...Ch. 9.SE - Prob. 32APCh. 9.SE - Prob. 33APCh. 9.SE - Propose structures for hydrocarbons that give the...Ch. 9.SE - Identify the reagents a-c in the following scheme:Ch. 9.SE - Prob. 36APCh. 9.SE - Prob. 37APCh. 9.SE - Prob. 38APCh. 9.SE - How would you carry out the following...Ch. 9.SE - Prob. 40APCh. 9.SE - Synthesize the following compounds using 1-butyne...Ch. 9.SE - Prob. 42APCh. 9.SE - Prob. 43APCh. 9.SE - Prob. 44APCh. 9.SE - Prob. 45APCh. 9.SE - A hydrocarbon of unknown structure has the formula...Ch. 9.SE - Compound A(C9H12) absorbed 3 equivalents of H2 on...Ch. 9.SE - Hydrocarbon A has the formula C12H8. It absorbs 8...Ch. 9.SE - Occasionally, a chemist might need to invert the...Ch. 9.SE - Prob. 50APCh. 9.SE - Prob. 51APCh. 9.SE - Prob. 52APCh. 9.SE - Prob. 53APCh. 9.SE - Prob. 54APCh. 9.SE - Prob. 55APCh. 9.SE - Prob. 56APCh. 9.SE - Prob. 57AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.arrow_forwardA certain hydrocarbon, C7H10, yields the two products shown below upon oxidative cleavage with KMnO4/H3O+ . Deduce the structure of the hydrocarbon.arrow_forwardPredict the product of the following epoxide ring opening and provide a mechanism for its formation. Be sure to include stereochemistry.arrow_forward
- Predict the major products of treating the following compound with hot, concentrated potassium permanganate, followed by acidification with dilute HCl. (a) p-xylenearrow_forwardPredict the products of the ff reactionSulfonation of acetophenone Oxidation of ethylbenzene with KMnO4arrow_forwardGive the expected products of lithium aluminum hydride reduction of the followingcompounds (followed by hydrolysis).(a) butyronitrile (b) N-cyclohexylacetamide (c) e@caprolactamarrow_forward
- PROPOSE A MECHANISM FOR THE REACTION OF PYRIDINE WITH H2SO4 AND HNO3. ACCOUNT FOR THE REGIOSELECTIVITY OF THE REACTION.arrow_forwardPredict the major product of E1 elimination of the following compounds and write a detailed mechanism for its formationarrow_forwardPredict the major products of treating the following compound with hot, concentrated potassium permanganate, followed by acidification with dilute HCl.(a) isopropylbenzenearrow_forward
- Which of the alkenes below is the most reactive towards acid-catalyzed hydration?arrow_forwardShow the product, including stereochemistry, of the reaction of the epoxide below with Na+ -CN, H2O.arrow_forwardWhen pyrrole is added to a dilute solution of D2SO4 in D2O, 2-deuteriopyrrole is formed. Propose a mechanism to account for the formation of this compound.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License