
a)

Interpretation:
How to convert 1-butyne in to butanone is to be shown.
Concept introduction:
Terminal
To show:
How to convert 1-butyne in to butanone.
b)
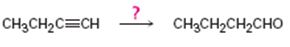
Interpretation:
How to convert 1-butyne in to butanal is to be shown.
Concept introduction:
Terminal alkynes when hydrated using hydroboration-oxidation reaction yield an enol with OH group attached to the terminal carbon as the reaction follows anti-Markovnikov regiochemistry. The enol obtained then tautomerizes to an
To show:
How to convert 1-butyne in to butanal is to be shown.
c)
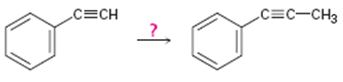
Interpretation:
How to convert ethynylbenzene in to propynylbenzene is to be shown.
Concept introduction:
Terminal alkynes are converted in to acetylides by treating with NaNH2 in liquid ammonia. The acetylides can be converted to higher alkynes by treating with
To show:
How to convert ethynylbenzene in to propynylbenzene.
d)
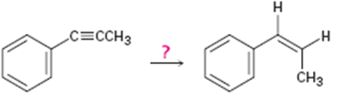
Interpretation:
How to convert propynylbenzene in to cis-1-propenylbenzene is to be shown.
Concept introduction:
Alkynes can be converted in to the corresponding
To show:
How to convert 1-propynylbenzene in to 1-propenylbenzene.
e)

Interpretation:
How to convert 1-butyne in to propanoic acid is to be shown.
Concept introduction:
Alkynes gets cleaved when treated with powerful oxidizing agents like KMnO4. Internal alkynes give carboxylic acids as products. Terminal alkynes give CO2 also along with a carboxylic acid.
To show:
How to convert 1-butyne in to propanoic acid.
f)
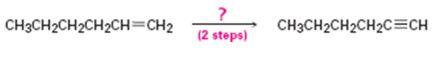
Interpretation:
How to convert 1-hexene in to 1-hexyne in two steps is to be shown.
Concept introduction:
Alkenes when treated with bromine yield a dibromo
To show:
How to convert 1-hexene in to 1-hexyne in two steps
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
