
a)
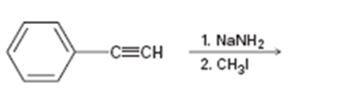
Interpretation:
The product of the two step process shown is to be given and complete electron-pushing mechanism for its formation also is to be provided.
Concept introduction:
The terminal
To propose:
The product of the two step process shown and to give a complete electron-pushing mechanism for its formation.
b)
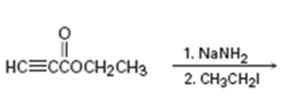
Interpretation:
The product of the two step process shown is to be given and complete electron-pushing mechanism for its formation also is to be provided.
Concept introduction:
The terminal alkynes being acidic form sodium alkynides when treated with bases like sodium amide. The alkynides when treated with alkyl halides yield the higher alkynes as the product. The alkynide ion being nucleophilic attacks the positively polarized carbon of C-X bond in alkyl halides displaces the halogen to yield the higher alkyne as the product.
To propose:
The product of the two step process shown and to give a complete electron-pushing mechanism for its formation.
c)
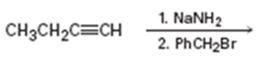
Interpretation:
The product of the two step process shown is to be given and complete electron-pushing mechanism for its formation also is to be provided.
Concept introduction:
The terminal alkynes being acidic form sodium alkynide when treated with bases like sodium amide. The alkynides when treated with alkyl halides yield the higher alkynes as the product. The alkynide ion being nucleophilic attacks the positively polarized carbon of C-X bond in alkyl halides displaces the halogen to yield the higher alkyne as the product.
To propose:
The product of the two step process shown and to give a complete electron-pushing mechanism for its formation.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- Provide the final product and mechanism for the following reactionsarrow_forwardPropose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each steparrow_forwardProvide the reagents to carry out the following reactionarrow_forward
- Provide the necessary sequence of reactions to generate the products below from their respective starting materials.arrow_forwardProvide reagents to carry out the following transformationsarrow_forwardProvide the reagent and mechanism for the transformation below. Explain in detail how the reaction conditions provide the given regioselectivity observed in the product.arrow_forward
- Describe a synthesis of the following molecules using the starting material and any additional reagents provided. There will be several steps involved.arrow_forwardCarry out the following reactions by providing the necessary reagent.arrow_forwardThe following are intermediate products in the stepwise synthesis of compound 1 from benzene. Give the correct sequence of reactions with the appropriate reagents that will lead to the correct intermediate products and final product.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





