
Concept explainers
Interpretation:
The reason for carbon dioxide to be nonpolar molecule even though its bonds are polar is to be explained.
Concept introduction:
There are two classes of molecules; polar and nonpolar. There are partial charges on atoms of a polar molecule. A polar molecule has a dipole as it has two poles; a positive end and a negative end.
Answer to Problem 3E
A polar molecule is a molecule that possesses partial charges on its atoms. In carbon dioxide molecules, the two dipole moments in CO2 balance each other, and there is no partial positive or negative charge on the molecule. So the overall molecule is nonpolar.
Explanation of Solution
In a carbon dioxide molecule, there is a double bond between carbon and oxygen atom. These bonds are polar. So there are two polar bonds in a molecule of CO2. Oxygen is more electronegative than carbon so both oxygen atoms pull the electrons away from the carbon atom in opposite directions. Both the dipoles in CO2 balance each other, and there is no partial positive or negative charge on the molecule. So the overall molecule is nonpolar.
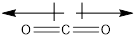
Carbon dioxide is a nonpolar molecule even though its bonds are polar as the two dipoles cancel out each other and there is no net charge on the molecule.
Chapter U2 Solutions
Living By Chemistry: First Edition Textbook
Additional Science Textbook Solutions
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach (4th Edition)
Organic Chemistry (8th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





