
Concept explainers
Give the IUPAC name for each compound.
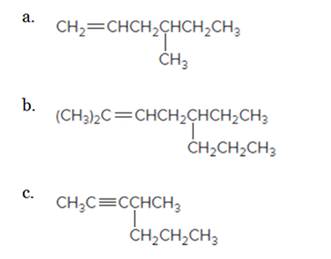
d.
e.
e.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Rank the following groups in order of decreasing priority. a. – COOH, – H, – NH2, – OH b. – H, – CH3, – Cl, – CH2CI c. -CH2CH3, -CH3, -H, -CH(CH3)2 d. – CH = CH2, – CH3, – C ≡ CH, – Harrow_forwardName the following: H H H H H – C – C – C = C - H H H Harrow_forward2,4-D is a synthetic auxin (a plant hormone) used in the control of broadleaf weeds, and one of the most widely used herbicides in the world. It is produced commercially by the following reaction: chloroacetic acid + dichlorophenol → hydrochloric acid + 2,4-D C2H3ClO2 + C6H4Cl2O → HCl + C8H6Cl2O3 How much chloroacetic acid does it take to manufacture 5.18 grams of 2,4-D?arrow_forward
- What is each compound’s systematic name? 1. (CH3)3CCH2CH2CH2CH(CH3)2 2. BrCH2CH2CH2CH2CH2NHCH2CH3 3.(CH3CH2)4C 4.CH3OCH2CH2CH2OCH3arrow_forward3. Classify the alcohols shown below as primary, secondary, or tertiary. A) secondary B) primary C) tertiary 1) CH3 - CH2 - OH 2) CH3 | CH3 - C - CH2 - OH | CH3 3) OH | CH3 - CH - CH3 4) 5) CH3 | CH3 - C - CH2 - CH3 | OH 4. Identify the product, if any, that would form in each of the following reactions. A) CH3 - CH3 B) CH3 - CH2 - CH3 C) OH | CH3 - C H - CH3 D) O CH3 - C - OH E) CH3 - CH2 - OH 1) O CH3 – C - H 2) O CH3 - C - CH3 + H2 3) O CH3 - C - H + H2arrow_forward2 Q: Consider the following compound: CH3-CH2-CO-CH3 a. suggest a method for increasing the number of carbons by one: b. suggest a method for decreasing the number of carbons by one:arrow_forward
- In the pic (yellow circle l). What is the above O means? Is it 5OH^- or 5 O^- H?arrow_forwardWhich of the following molecules has only single bonds. A. CHCHCH3 B. CH2CHCH3 C. CH3CH2CCH D. CH3CH3 E. CH2CH2 Which of the following molecules has a carbon-to-carbon double bond? A. CH3CCH B. CHCH C. CH3CH3 D. CH3CH2CH3 E. CH2CHCH3arrow_forwardIf the computed Kc of an unknown compound is equal to 1.3, where does chemical equilibrium lie? D. left to right C. right B. left A. centerarrow_forward
- the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (h).arrow_forwardPlace the following compunds in order from lowest to highest boling points. Explain why you have ranked each compound C6H14 CH3OH H2O C3H8arrow_forwardRank the following compounds according to increasing positive character of the carbon atom. least positive C Most positive C CH3F CH3OH CH3Li CH3I CH3CH3 CH3NH2arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

