
Concept explainers
(a)
Interpretation:
Each pair from the following structures should be identified as constitutional isomers, stereoisomers or identical.
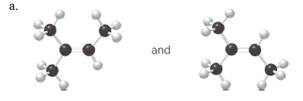
Concept Introduction:
Isomers are the different molecules which have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
(b)
Interpretation:
Each pair from the following structures should be identified as constitutional isomers, stereo isomers or identical.
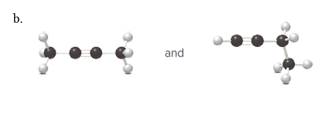
Concept Introduction:
Isomers are the different molecules which have the same molecular formula but differ in molecular arrangement in the space. There are several groups of isomers.
Stereoisomers are the form of isomers which have different three-dimensional orientations in the space.
Constitutional isomers are the form of isomers which have atoms bonded to each other in different ways.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How do the products formed from each reactant compare—identical compounds, stereoisomers, or constitutional isomers?arrow_forwardClassify each pair of compounds as either identical, constitutional isomers, stereoisomers, or not isomers.arrow_forwardLabel each pair as constitutional isomers, stereoisomers, or not isomers of each.arrow_forward
- Classify each pair of compounds as constitutional isomers, stereoisomers, identical molecules, or not isomers of each attached otherarrow_forwardClassify each pair of compounds as constitutional isomers, stereoisomers, identical molecules or unrelated molecules:arrow_forwardLabel following pairs of molecules as being either same same structure, completely different, constitutional isomers, or stereoisomers.arrow_forward
- 10. What is the relationship between the following two compounds? (see attached screenshot). a. constitutional isomers b. stereoisomers c. identical d. not isomers; different compounds entirelyarrow_forwardClassify each pair of compounds as constitutional isomers or stereoisomers.arrow_forwardTell whether the following pairs of compounds are identical constitutional isomers, or stereoisomers.arrow_forward
- Do constitution isomers have the same boiling point or melting point?arrow_forwardIllustrate examples of both types of isomers ? a. constitutional isomers b. stereoisomersarrow_forwardWhich statements are true about constitutional isomers? (a) They have the same molecular formula. (b) They have the same molecular weight. (c) They have the same order of attachment of atoms. (d) They have the same physical propertiesarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
