
Concept explainers
(a)
Interpretation:
Each carbon in the following molecule should be labeled as tetrahedral, trigonal planar or linear:
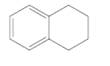
Concept Introduction:
Valence shell electron pair repulsion (VSEPR) theory is used to determine the shape. VSEPR is based on the fact that electron pairs repel each other; thus, when groups (a group is either an atom or a lone pair of electrons) are as far away from each other as possible then they are in most stable arrangement.
(b)
Interpretation:
Each carbon in the following molecule should be labeled as tetrahedral, trigonal planar or linear:
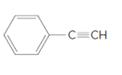
Concept Introduction:
Valence shell electron pair repulsion (VSEPR) theory is used to determine the shape. VSEPR is based on the fact that electron pairs repel each other; thus, when groups (a group is either an atom or a lone pair of electrons) are as far away from each other as possible then they are in most stable arrangement.
(c)
Interpretation:
Each carbon in the following molecule should be labeled as tetrahedral, trigonal planar or linear:
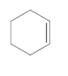
Concept Introduction:
Valence shell electron pair repulsion (VSEPR) theory is used to determine the shape. VSEPR is based on the fact that electron pairs repel each other; thus, when groups (a group is either an atom or a lone pair of electrons) are as far away from each other as possible then they are in most stable arrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
General, Organic, and Biological Chemistry - 4th edition
- (a)({)-hexane-3,4-diol, label each structure you have drawn as chiral or achiral.arrow_forwardConvert each compound to a skeletal structure. a. CH3(CH2)7CH3 window open b. 1,1−diethylcyclohexane draw structure ... c. (CH3CH2)2CHCH2CH2CH3 draw structure ...arrow_forward4-ethyl-5,5-dimethyl-3-isopropyloctane whay does this structure look like with all the C's and H's?arrow_forward
- 1. Draw the structure for each compound. a.(3R)-3-methylhexane b. (3R,5S,6R)-5-ethyl-3,6-dimethylnonanearrow_forwardGive the structure corresponding to each IUPAC name. d. 1-ethyl-2,3dimethylcyclopentane e. 1,5-dimethylcyclohexanearrow_forwardClassify each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°).(a) butane (b) isobutane (c) 2-methylbutane(d) cyclohexane (e) norbornane (bicyclo[2.2.1]heptane)arrow_forward
- Draw the Kekule Structure with lone pairs, the type of organic reaction (addition, elimination, substitution, and rearrangement reaction), and draw the move of electrons of the following: 1. H2C=CH2 + H2 --> CH3CH3arrow_forwardDraw three constitutional isomers having molecular formula C 7H 14 that contain a fi ve-membered ring and two CH 3 groups bonded to the ring. Give the IUPAC name for each isomerarrow_forwardLabel each carbon and hydrogen atoms of the following molecules as primary 1°, secondary 2°, tertiary 3°, or quaternary 4: 1. Neopentane 2. 2,2,4-trimethylpentane 3. 2-cyclopropylpentanearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


