
(a)
Interpretation:
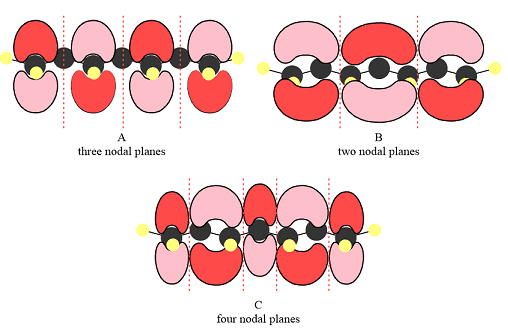
The number of nodal planes perpendicular to the bonding axes of each of the three
Concept introduction:
Overlap of atomic orbitals (AOs) can be thought of as wave interference. It can be constructive or destructive. Constructive interference increases the electron density between two nuclei (an antinode) and results in a molecular orbital (MO) of lower energy. The phases of overlapping orbitals are the same in this case. Destructive interference decreases the electron density between two nuclei and results in an MO of higher energy. The phases of such AOs are the opposite. Since the electron density between two nuclei decreases, there is a node (or a nodal plane) between the two atoms.
Answer to Problem 14.26P
The number of nodal planes is three for MO A, two for MO B, and four for MO C.
The order of increasing energy of the three MOs is
Explanation of Solution
The phases of the wave function of the two AOs on either side of a nodal plane are opposite each other. This results in destructive interference reducing the electron density to zero at the nodal plane.
Therefore, the nodal planes in the three MOs are:
The energy of the MO increases with the number of nodes. Therefore, the order of increasing energy of the three orbitals is
When atomic orbitals with different phases overlap, a node (zero electron density) is formed at the center, increasing the energy of the MO.
(b)
Interpretation:
The p orbital AO contributions on each carbon atom that would give rise to each MO are to be drawn.
Concept introduction:
The p orbitals that contribute to
Answer to Problem 14.26P
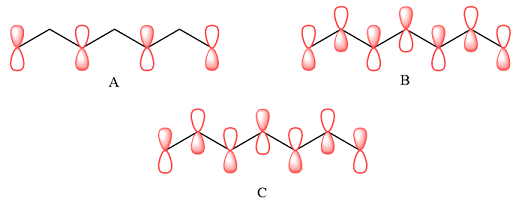
The p orbitals that contribute to each of the three MOs are
Explanation of Solution
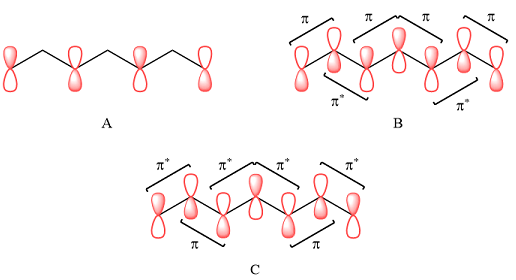
In case of A, the MOs shown are present on alternate carbon atoms. This means they do not overlap and are non-bonding MOs. The corresponding AOs are p orbital on alternate carbon atoms. There is no contribution from atoms that lie in the nodal planes. The p orbitals on either side of each nodal plane have opposite phases.
Therefore, the p orbitals that contribute to MOs shown in A are
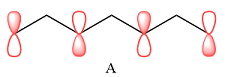
There are no bonding or antibonding interactions in this case.
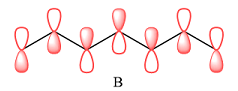
In case of B, there are three bonding MOs. The p orbitals of C1 and C2 (from left) overlap to produce a bonding MO, therefore, they must be in phase. The p orbitals of C3, C4, and C5 overlap constructively to produce a three-center bonding MO. The p orbitals of C6 and C7 overlap to produce another bonding MO. There are two nodal planes, one between C2 and C3, and the second between C5 and C6. This means that the phases of the MOs, and therefore of the contributing AOs on the two sides of each nodal plane must be opposite.
Therefore, the p orbitals contributing to the MOs as shown in B are
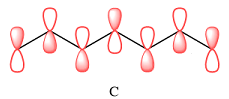
In case of C, there are three nodal planes, between C1-C2, between C3-C4, between C4-C5, and between C6-C7. The phases of the orbitals on either side of each nodal plane must be opposite. There are only two bonding MO interactions, the first between C2 and C3 and the second between C5 and C6, therefore, these pairs must be of the same phase.
Therefore, p orbitals contributing to the MOs as shown in C are
The p orbitals contributing to each MO are determined on the basis of the phases and the presence of an adjacent nodal plane.
(c)
Interpretation:
Each internuclear region is to be identified as having a bonding or an antibonding type of interaction.
Concept introduction:
A bonding interaction arises when the phases of the interacting AOs are the same. This increases the electron density between the two nuclei and lowers the energy of the MO. An antibonding interaction arises when the phases of the interacting AOs are different. This decreases the electron density between the two nuclei to zero at the center and increases the energy of the MO.
Answer to Problem 14.26P
The bonding (
Explanation of Solution
A bonding interaction requires p orbitals of the same phase on adjacent atoms. An antibonding interaction requires the interacting p orbitals to be of opposite phases. An antibonding interaction leads to a nodal plane between the two atoms.
Therefore, in A, there are no bonding or antibonding interactions as the AOs are not on adjacent carbon atoms. In case of B, there are bonding interactions between p AOs of C1 and C2 (from left), between C3, C4, and C5, and between C6 and C7. There are antibonding interactions between C2 and C3 and between C5 and C6.
In case of C, there are two bonding interactions, between C2 and C3, and between C5 and C6. The remaining four interactions are antibonding.

The type of interaction is determined on the basis of the phases of the interacting AOs.
(d)
Interpretation:
Whether each MO is overall bonding, nonbonding or antibonding is to be determined on the basis of the answer to part (c).
Concept introduction:
If the number of bonding interactions are more than the number of antibonding interactions, the MO is overall bonding. If the number of antibonding interactions is more than that of bonding interactions, the MO is overall antibonding. If the numbers are equal or there are no interactions, the MO is overall nonbonding.
Answer to Problem 14.26P
The MO shown in A is overall nonbonding. MO B is overall bonding. MO C is overall antibonding.
Explanation of Solution
The contributing p AOs are on alternate carbon atoms, and therefore, not in a position to interact. Therefore, there are no bonding or antibonding interactions in this case. Therefore, the MO shown in A is overall nonbonding.
In case of B, there are four bonding and two antibonding interactions. Effectively, this means the MO is overall bonding.
In case of C, there are only two bonding interactions but four antibonding interations. Therefore, this MO is overall antibonding.
The overall character of a multicenter MO is determined by the numbers of bonding and antibonding interactions.
Want to see more full solutions like this?
Chapter 14 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- For each of the following molecules, construct the MOs from the 2pz atomic orbitals perpendicular to the plane of the carbon atoms. (a) Cyclobutadiene HC (b) Allyl radical Indicate which, if any, of these orbitals have identical energies from symmetry considerations. Show the number of electrons occupying each MO in the ground state, and indicate whether either or both of the molecules are paramagnetic. Assume that the C atoms in the allyl radical are all sp2 hybridized.arrow_forwardThe FEMO theory of conjugated molecules is crude and marginally better results are obtained with simple Huckel theory. (a) For a linear conjugated polyene with each of N carbon atoms contributing an electron in a 2porbital, the energies Ek of the resulting π molecular orbitals are given by: Ek = α + 2β cos kπ/N+1 k = 1,2,3, ... N Use this expression to estimate the resonance integral β for the series consisting of ethene, butadiene, hexatriene, and octatetraene given that ult raviolet absorptions from the HOMO, which is a bonding π orbital, to the LUMO, which is an antibonding π* orbital, occur at 61 500, 46 080, 39 750,and 32 900 cm- 1, respectively. (b) Calculate the π-electron delocalization energy, Edelcc = Eπ -n(α + β). of octatetraene, where Eπ, is the total π-electron binding energy and n is the total number of π-electrons.arrow_forwardHow would you draw a molecular orbital diagram of butadiene showing the energies and occupancy ofthe orbitals in the ground state? How would you estimate the total pi binding energy of the molecule?arrow_forward
- Construct the molecular-orbital energy level diagramsof (a) ethene and (b) ethyne on the basis that the moleculesare formed from the appropriately hybridized CH2 or CH fragmentsarrow_forwardHomolytic bond breaking happens when there is an equal separation of electron in the presence of UV. true or false?arrow_forwardFor monocyclic conjugated polyenes (such as cyclobutadiene and benzene) with each of N carbon atoms contributing an electron in a 2p orbital, Huckel theory gives the fol lowing expression for the energies Ek of the resulting π molecular orbitals Calculate the energies of the 1t molecular orbitals of benzene and cyclooctatetraene. Comment on the presenceor absence of degenerate energy levels. (b) Ca lcu late and compare the delocalization energies of benzene and hexatriene using the expression from the preceding problem. What do you conclude from your results? (c) Calculate and compare the delocalization energies of cyclooctatetraeneand (linear) octatetraene. Are your conclusions for th is pair of molecules the same as for the pair of molecules investigated in part (b)?arrow_forward
- Solve for the delocalization and overall electronic energy for C4H4 (considering a single p orbital on each carbon) ? How does this compare to benzene?arrow_forwarddelta S of 2CH3CHO +3O2 -> 2CO2 + 4H2Oarrow_forwardWhich MOs are the HOMO and the LUMO in the ground state? for the MOs of 1,3,5-hexatrienearrow_forward
- In the reaction of CN- + H + → HCN; 1) Which boundary orbitals are used? Draw a diagram and explain according to MOT. 2) Explain the orbital overlaps of the product according to VBT. 3) Determine the point group of the product.arrow_forwardFor each molecule or molecular ion below, calculate the bond order: N2, N2-, O2 and O2+. In this calculation, show which specifically bonding and anti-bonding molecular orbitals were used in the calculation. Please label molecular orbitals with appropriate “g” (“gerade”) and “u” (“ungerade”) subscripts.arrow_forwardSet up and solve the secular determinants for (a) cyclobutadiene in the Hückel p-electron scheme (considering a single p orbital on each carbon); find and order the energy levels and molecular orbitals, and estimate the delocalization energyarrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




