
(a)
Interpretation:
Fischer projection for a given molecule needs to be determined.
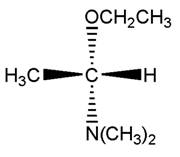
Concept Introduction:
Fischer Projection is a method of drawing 3-D structures of organic molecules in 2D nature. In this method, all non-terminal bonds are depicted as horizontal or vertical lines.
For a simple molecule consists of one chiral center with four different groups attached (e.g. CHClBrI), the Fischer projection method uses a cross formula to draw the two bonds in the plane. In the Fischer projection, both horizontal bonds come forward (drawn on wedges) and both vertical bonds go behind (on dashed lines).
(b)
Interpretation:
Fischer projection for a given molecule needs to be determined.
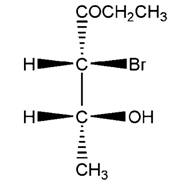
Concept Introduction:
Fischer Projection is a method of drawing 3-D structures of organic molecules in 2D nature. In this method, all non-terminal bonds are depicted as horizontal or vertical lines.
For a simple molecule consists of one chiral center with four different groups attached (e.g. CHClBrI), the Fischer projection method uses a cross formula to draw the two bonds in the plane. In the Fischer projection, both horizontal bonds come forward (drawn on wedges) and both vertical bonds go behind (on dashed lines).
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Convert each compound to a Fischer projection, and label each stereogenic center as R or S.arrow_forwardRe-draw each Fischer projection formula using wedges and dashed wedgesfor the stereogenic center, and label the center as R or S.arrow_forwardDrawing sugars to and from fischer projectionsarrow_forward
- Draw a Fischer projection for each compound. Remember that the cross represents an asymmetric carbon atom, and the carbon chain should be along the vertical, with the IUPAC numbering from top to bottom. (S)-1,2-dibromobutanearrow_forwardFor those that have only one stereocenter draw Fischer projection(s) of the S-stereoisomer placing the 4 and 2 positions on the vertical line.arrow_forwardDraw the Fischer projection from the Hawthorne projection shown below.arrow_forward
- Draw a Fischer projection for each compound. Remember that the cross represents an asymmetric carbon atom, and the carbon chain should be along the vertical, with the IUPAC numbering from top to bottom.(R)-glyceraldehyde,arrow_forwardDraw the Fischer projection for structure I. Circle each chiral group in structure II.arrow_forwardClassify each compound as IDENTICAL to A or an ENANTIOMER to A.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
