
Concept explainers
(a)
Interpretation:
The chiral center in the following compound should be located.
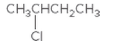
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and the compound is known as a chiral compound.
(b)
Interpretation:
The chiral center in the following compound should be located.
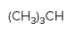
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and the compound is known as a chiral compound.
(c)
Interpretation:
The chiral center in the following compound should be located.
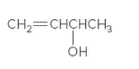
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and compound is known as a chiral compound.
(d)
Interpretation:
The chiral center in the following compound should be located.
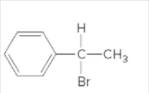
Concept Introduction:
Compounds in which an atom is attached to four different groups of atoms are known as chiral center and compound is known as a chiral compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Label the chirality center (if one exists) in each compound. A compound may contain zero or one chirality center.arrow_forward1. Isomerism and Stereochemistry A. Identify the configuration of the double bond. B. Redraw the structure and identify the chiral centers using an asterisk. C. Redraw the structure with all the chiral centers in R configuration.arrow_forwardHow many chirality centers are in each compoundarrow_forward
- What are diastereomers? a. Constitutional isomers. b. Molecules with only one chiral center. c. Stereoisomers that are not mirror images of one another and are non-superimposable on one another. d. Stereoisomers that are mirror images of one another.arrow_forwardEphedrine (2‑methylamino‑1‑phenylpropanol) is a drug that is used as a decongestant and bronchodilator to treat, among other conditions, the congestion due to the common cold. Place an asterisk (*) next to the chirality center(s) in the ephedrine molecule.arrow_forward2. Draw one regioisomer and one stereoisomer and assign R/S for chiral centers. Galantamine Structurearrow_forward
- Because there is usually slow interconversion between the two isomeric forms at room temperature. Because there is usually rapid interconversion between the two isomeric forms at room temperature. Because chirality only exists with the tetrahedral carbon atoms. Because four bonds a are needed to define a stereogenic center.arrow_forwardConvert each three-dimensional representation into a Fischer projection.arrow_forwardUse an asterisk to label each of the chirality centers in the following moleculearrow_forward
- 1. a.) Put asterisk on chiral centers b.) label each chiral center as R or S. c.) Draw the enantiomer d.) Draw one diastereomerarrow_forwardA compound with two chirality centers is shown (2S,3R)-2-bromo-3-chlorobutane. Convert the structure to the wedge and dash structurearrow_forwardIdentify the configuration of each chiral center in the following compound Chiral center locations: Configuration(s):arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

