
Concept explainers
(a)
Interpretation:
The structure of the alkyl bromide of molecular formula
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image.
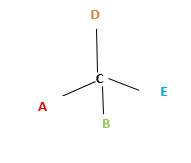
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different
(b)
Interpretation:
The structure of the
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image.
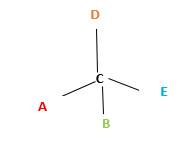
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different functional groups.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 3.A carbon atom that is bonded to four different groups is a/an A.achiral center B.chirality center C.symmetrical center D.symmetrical centerarrow_forwardWhat is the significance of a reaction that occurs in 94% enantiomeric excess? A. The product contains 94% of one enantiomer and 6% of other products B. The product contains 94% of one enantiomer and 6% of another enantiomer C. The product contains an enantiomer that is 94% pure. D. Product contains 97% of one enantiomer and 3% of another enantiomerarrow_forwardLabel the chirality center (if one exists) in each compound. A compound may contain zero or one chirality center.arrow_forward
- A(n) ________ is an achiral compound that contains chiral centers but is superimposable on its mirror image. A) constitutional isomers B) conformational isomers C) enantiomers D) diastereomers E) meso compoundsarrow_forwardWhat is the relationship between the two molecules shown below? A) enantiomers B) diastereomers C) conformational isomers D) constitutional isomers E) identicalarrow_forwardWhich statement is correct? a. Testosterone and nortestosterone possess a ketone at C-3 conjugated to a single bond at C-4 and a hydroxyl at C-17b. b. Testosterone and nortestosterone possess a ketone at C-4 conjugated to a double bond at C-3 and a hydroxyl at C-17b. c. Testosterone and nortestosterone possess a ketone at C-3 conjugated to a double bond at C-4 and a hydroxyl at C-17b. d. The obviously wrong answer!.arrow_forward
- Determine the relationship between the two drawings: A. Enantiomers B. Same compound C. Constitutional isomers D. Diastereomersarrow_forwardDraw the products formed when cembrene A is treated with O3 followed by CH3SCH3. Label each product as chiral or achiralarrow_forwardDraw compounds that fit the following descriptions: (a) A chiral alcohol with four carbons (b) A chiral carboxylic acid with the formula C5H10O2 (c) A compound with two chirality centers (d) A chiral aldehyde with the formula C3H5BrOarrow_forward
- Two diastereomeric sets of enantiomers, A/B and C/D, exist for 3-bromo-2-butanol. When enantiomer A or B is treated with HBr, only racemic 2,3-dibromobutane is formed; no meso isomer is formed. When enantiomer C or D is treated with HBr, only meso 2,3-dibromobutane is formed; no racemic 2,3-dibromobutane is formed. Account for these observations.arrow_forwardThere are nine constitutional isomers of molecular formula C7H16. 1) For the two chiral constitutional isomers, draw in Fischer projection formula the enantiomers of each. 2) Name, according to IUPAC standards, each of the enantiomers you drew in #4 above.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning



