
Concept explainers
(a)
Interpretation:
The relationship between the given pair of molecules needs to be explained.

Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. Molecules that do not have any chiral centers are called achiral. Identical molecules do not have any chiral centers; therefore, they are achiral.
When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
When a molecule has more than one chiral center, another class of stereoisomers can be defined: Diastereomers, are the stereoisomers, which are not mirror images of each other.
(b)
Interpretation:
The relationship between the given pair of molecules needs to be explained.

Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. Molecules that do not have any chiral centers are called achiral. Identical molecules do not have any chiral centers; therefore, they are achiral.
When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
When a molecule has more than one chiral center, another class of stereoisomers can be defined: Diastereomers, are the stereoisomers, which are not mirror images of each other.
(c)
Interpretation:
The relationship between the given pair of molecules needs to be explained.

Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. Molecules that do not have any chiral centers are called achiral. Identical molecules do not have any chiral centers; therefore, they are achiral.
When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
When a molecule has more than one chiral center, another class of stereoisomers can be defined: Diastereomers, are the stereoisomers, which are not mirror images of each other.
(d)
Interpretation:
The relationship between the given pair of molecules needs to be explained.
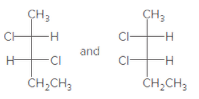
Concept Introduction:
Identical molecules are the ones with no isomers, neither constitutional isomers nor stereoisomers. Identical molecules have the same structural arrangement of atoms and the same three-dimensional arrangement.
Isomers are the molecules with the same formula but either with different structural connectivity (constitutional isomers) or different three-dimensional arrangement (stereoisomers).
A carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. Molecules that do not have any chiral centers are called achiral. Identical molecules do not have any chiral centers; therefore, they are achiral.
When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
When a molecule has more than one chiral center, another class of stereoisomers can be defined: Diastereomers, are the stereoisomers, which are not mirror images of each other.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- For each pair of compounds, please explain if they are identical. comstitutional isomers, enantiomers, or diastereomers.arrow_forwardDetermine each as enantiomers, diastereomers, constitutional isomers, or the same molecule.arrow_forwardHow are the compounds in attached pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other?arrow_forward
- What are diastereomers? a. Constitutional isomers. b. Molecules with only one chiral center. c. Stereoisomers that are not mirror images of one another and are non-superimposable on one another. d. Stereoisomers that are mirror images of one another.arrow_forwardState how each pair of compounds is related. Are they enantiomers, diastereomers, constitutional isomers, or identical?arrow_forwardAre they the same molecule, not even isomers, Constitutional Isomers, Diastereomers, or Enantiomers?arrow_forward
- For each pair, indicate whether they are constitutional isomers, enantiomers, diastereoisomers, or identical molecules. A BDEFarrow_forwardwhat is the relationship between these compounds, constitutional isomer, enantiomer, diastereomer, same compound?arrow_forwardDetermine whether each pair of molecules represent: (a) identical compounds; (b) constitutional (structural) isomers, (c) enantiomers, or (d) diastereomers. Thank you.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



