
(a)
Interpretation:
The following pair of molecules should be classified as stereoisomers, constitutional isomers, or identical molecules:
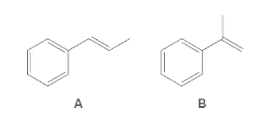
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms or group of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
(b)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:
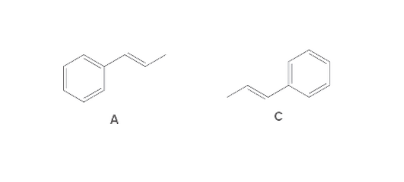
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
(c)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:
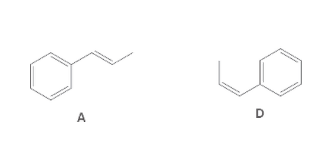
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Classify attached pair of compounds as constitutional isomers or stereoisomers ?arrow_forwardHow are the compounds in attached pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other??arrow_forwardExplain the similarities and differences between constitutional isomers and stereoisomersarrow_forward
- How is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules, constitutional isomers, or not isomers of each other.arrow_forwardCompounds E and F are two isomers of 2,3-dibromopentane drawn in staggered conformations. Which compounds (A–D) in Figure 5.8 are identical to E and F?arrow_forwardClassify each attached pair of compounds as stereoisomers or conformations: (a) X and Y; (b) X and Z.arrow_forward
- Consider the attached tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each sixmembered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.arrow_forwardDetermine whether each pair of molecules represent: (a) identical compounds; (b) constitutional (structural) isomers, (c) enantiomers, or (d) diastereomers. Thank you.arrow_forwardClassify each pair of compounds as constitutional isomers or stereoisomers.arrow_forward
- Drawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D? b.Label each compound as chiral or achiral. c.Which compounds alone would be optically active? d.Which compounds have a plane of symmetry? e.How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D? f.Which of the compounds are meso compounds? g.Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?arrow_forwardLabel following pairs of molecules as being either same same structure, completely different, constitutional isomers, or stereoisomers.arrow_forward(a) Draw a skeletal structure of the anabolic steroid methenolone from the following description. Methenolone contains the tetracyclic steroid skeleton with a carbonyl group at C3, a hydroxyl at C17, a double bond between C1 and C2, and methyl groups bonded to C1, C10, and C13. (b) Add wedges and dashed wedges for all stereogenic centers with thefollowing information: the configuration at C10 is R, the configuration at C13 is S, the configuration at C17 is S, and all substituents at ring fusions are trans to each other. (c) Draw the structure of Primobolan, the product formed when methenolone is treated with CH3(CH2)5COCl and pyridine. Primobolan is an anabolic steroid that can be taken orally or by injection and has been used illegally by well-known Major League Baseball players.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
