
Concept explainers
(a)
Interpretation:
Anomer of the aldohexose should be converted to its Haworth projection.
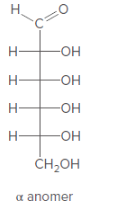
Concept Introduction:
Haworth formula indicates the connectivity of atoms in a ring structure. The linear structure should be used in the conversion.
(b)
Interpretation:
Anomer of the aldohexose should be converted to its Haworth projection.
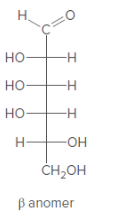
Concept Introduction:
Haworth formula indicates the connectivity of atoms in a ring structure. The linear structure should be used in the conversion.
(c)
Interpretation:
Anomer of the aldohexose should be converted to its Haworth projection.
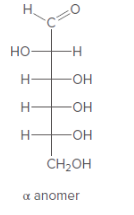
Concept Introduction:
Haworth formula indicates the connectivity of atoms in a ring structure. The linear structure should be used in the conversion.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Answer the following questions about monosaccharide A.a. Draw the α anomer of A in a Haworth projection.b. Draw the β anomer of A in a three-dimensional representation using a chair conformation.c. What two aldoses yield A in a Wohl degradation?d. What product is formed when A undergoes a Wohl degradation?e. What product is formed when A reacts with Ag2O in NH4OH?arrow_forwardLabel each Haworth projection as an α or β anomer and convert the Haworth projection to a sixmembered ring with wedges and dashed wedges.arrow_forwardLabel the four stereogenic centers in sorbitol as R or S. (see attached file)arrow_forward
- Which D-aldopentose is oxidized to an optically active aldaric acid and undergoes the Wohl degradation to yield a D-aldotetrose that is oxidized to an optically active aldaric acid?arrow_forward5-Deoxyglucose will close to a?arrow_forwardThe following isomerization reaction, drawn using D-glucose as starting material, occurs with all aldohexoses in the presence of base. Draw a stepwise mechanism that illustrates how each compound is formed.arrow_forward
- Treatment with sodium borohydride converts aldose A to an optically inactive alditol. Wohl degradation of A forms B, whose alditol is optically inactive. Wohl degradation of B forms d-glyceraldehyde. Identify A and B.arrow_forwardD-Arabinose can exist in both pyranose and furanose forms. a. Draw the α and β anomers of D-arabinofuranose. b. Draw the α and β anomers of D-arabinopyranose.arrow_forwardDraw the two possible Haworth structures for each the following sugars in their cyclized form. Be sure to label each as an ? or a ? anomer.arrow_forward
- Label attached Haworth projection as an α or β anomer, and convert the Haworth projection to a six-membered ring with wedges and dashed wedges.arrow_forwardConvert each Haworth projection to its acyclic formarrow_forwardAn oligosaccharide isolated from an organism is found tocontain two glucose residues and one galactose residue.Exhaustive methylation followed by hydrolysis producedtwo glucoses with methoxy groups at positions 2, 3,and 6 and galactose with methoxy groups at positions2, 3, 4, and 6. What is the structure of the originaloligosaccharide?arrow_forward
