
Concept explainers
(a)
Interpretation: To identify the substances among malate, oxaloacetate, fumarate,
Concept introduction: There are a number of
Polyfunctional carboxylate ions act as a substrate in the metabolic pathways and can be divided into two parts depending upon the parent
The first class consists of malate, oxaloacetate and fumarate ion which are derivative of succinic acid.
The chemical structures for carboxylate ion derived from succinic acid are as follows:
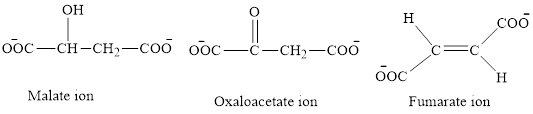
The second class consists of
The chemical structures for carboxylate ion derived from glutaric acid are as follows:
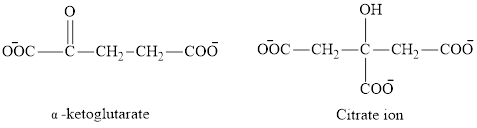
(b)
Interpretation: To identify the substances malate, oxaloacetate, fumarate,
Concept introduction: There are a number of metabolic reaction that occur in the human body for completion of functions. During these metabolic reactions, the various metabolic intermediates are formed for the short time to complete the reactions. Polyfunctional carboxylate ions are also the metabolic intermediate formed during metabolic pathways.
Polyfunctional carboxylate ions act as a substrate in the metabolic pathways and can be divided into two parts depending upon the parent carboxylic acid.
The first class consists of malate, oxaloacetate and fumarate ion which are derivative of succinic acid.
The chemical structures for carboxylate ion derived from succinic acid are as follows:

The second class consists of
The chemical structures for carboxylate ion derived from glutaric acid are as follows:

(c)
Interpretation: To identify the substances malate, oxaloacetate, fumarate,
Concept introduction: There are a number of metabolic reaction that occur in the human body for completion of functions. During these metabolic reactions, the various metabolic intermediates are formed for the short time to complete the reactions. Polyfunctional carboxylate ions are also the metabolic intermediate formed during metabolic pathways.
Polyfunctional carboxylate ions act as a substrate in the metabolic pathways and can be divided into two parts depending upon the parent carboxylic acid.
The first class consists of malate, oxaloacetate and fumarate ion which are derivative of succinic acid.
The chemical structures for carboxylate ion derived from succinic acid are as follows:

The second class consists of
The chemical structures for carboxylate ion derived from glutaric acid are as follows:

(d)
Interpretation: To identify the substances malate, oxaloacetate, fumarate,
Concept introduction: There are a number of metabolic reaction that occur in the human body for completion of functions. During these metabolic reactions, the various metabolic intermediates are formed for the short time to complete the reactions. Polyfunctional carboxylate ions are also the metabolic intermediate formed during metabolic pathways.
Polyfunctional carboxylate ions act as a substrate in the metabolic pathways and can be divided into two parts depending upon the parent carboxylic acid.
The first class consists of malate, oxaloacetate and fumarate ion which are derivative of succinic acid.
The chemical structures for carboxylate ion derived from succinic acid are as follows:

The second class consists of
The chemical structures for carboxylate ion derived from glutaric acid are as follows:

Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Classify each of the following sugar pairs as enantiomers, diastereomers, epimers, or an aldose–ketose pair. a. D-erythrose and D-threose b. D-glucose and D-mannose c. D-ribose and L-ribose d. D-allose and D-galactose e. D-glyceraldehyde and dihydroxyacetonearrow_forwardListed below are descriptions that may belong to amylose, amylopectin, both, or it may not belong to either. Write A if it describes amylose, B if it describes amylopectin, AB if the statement applies to both, or O if it doesn’t apply to either. 1. Its monosaccharides are bound by at least one α-1,4-glycosidic bond. 2. It contains α-1,6-glycosidic bonds. 3. It can be broken down by the enzyme α-amylase. 4. A polysaccharide made up of gulose. 5. It can form a double helix.arrow_forwardDefine the following terms: a. ganglioside b. sphingolipidoses c. isoprenoid d. terpene e. mixed terpenoidarrow_forward
- Name the following compound: HO HO H H OH H. OH но он ÓH H HO, а. Ketose b. Maltose c. Sucrose d. Aldosearrow_forwardWhich of the following is/are NOT (an) essential fatty acids? A. arachidonic acid B. linoleic acid C. palmitic D. oleic acidarrow_forwardDefine the following terms: a. lactone b. aldaric acid c. aldonic acid d. uronic acid e. epimerizationarrow_forward
- Which of the following is/are (a) ketopentose? A. threose B. xylulose C. arabinose D. mannosearrow_forwardLook at the structure of stearic acid as well as oleic acid and answer the following questions: a.Write which of these two is saturated fatty acid. b.Which of these two has a higher melting points. c.Which of these two will change the color of bromine water to clear. d.Name the unsaturated fatty acid using both delta and Omega nomenclaturearrow_forwardWhat is the common feature between an aldo- hexose and a ketohexose: A The number of secondary alcohol functions B The number of carbonyl groups C The number of asymmetric carbons D The number of primary alcohol functions E The nature of their carbonyl grouparrow_forward
- Which of the following statements about trans-fatty acids is/are FALSE? a.Healthier than saturated FA. b.Formed from the hydrogenation of C=C. c.May be present in butter substitutes. d.Properties are similar to saturated FA.arrow_forwardWhich of the following is not a saturated fatty acid? A. Linoleic acid B. Caproic acid C. Myristic acid D. Palmitic acidarrow_forwardSorbitol is a sweetener often substituted for cane sugar, because it is better tolerated by diabetics. It is also an intermediate in the commercial synthesis of vitamin C. Sorbitol is prepared by high-pressure hydrogenation of glucose over a nickel catalyst. What is the structure (including stereochemistry) of sorbitol?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





