
Concept explainers
(a)
Interpretation:
The pKa value for a protonated
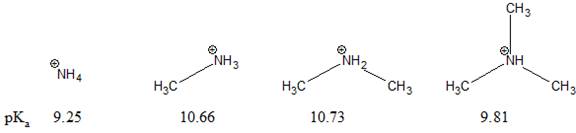
Concept introduction:
The stability of a charged species depends on the groups attached to the positively charged atom. The electron withdrawing group increases the concentration of positive charge on the nearby atom. While, the electron donating group decreases the concentration of positive charge on the nearby atom. This increases the stability of nearby positively charged atom. In general, cations are stabilized by electron-donating groups near the positive charge.
(b)
Interpretation:
The pKa value for a protonated amine (R3NH+) depends on the number of alkyl groups attached to N, as shown below. This order disagrees with what we would predict using charge stability. The reason for the discrepancy is to be explained.
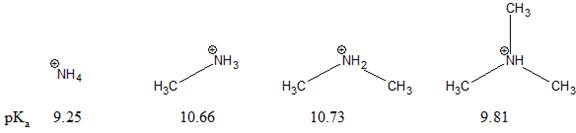
Concept introduction:
The stability of a charged species decreases as the effective electronegativity of an atom bearing a positive charge increases. The electron donating group decreases the concentration of positive charge on the nearby atom. This increases the stability of nearby positively charged atom. In general, cations are stabilized by electron-donating groups near the positive charge. Bulky group around the cation, reduces the solvation process. This decreases the stability of the ion and increases acidity.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- explain in great detail why the pKa values are what they are and compare them among the three compounds. The conjugate acids that are protonated are drawn in red for each compound. Which one based on the pKa is most acidic and what does that tell you about how basic their lone pairs are?arrow_forwardWhich is a stronger acid and has the most stable conjugate base? Estimate the pKA values for both compounds.arrow_forwardThe following compound is derived from one of the twenty mammaliam "amino acids". It is the ethyl ester of arginine . If you offer a proton (H+) to this molecule, what is the expected conjugate acid that is formed. Draw its structure, thank you!arrow_forward
- Given that Kb for ammonia is 1.8 * 10-5 and that for hydroxylamine is 1.1 * 10-8, which is the stronger base?arrow_forwardwhich of the following explain the relative acidity of the indicated protons?arrow_forwardWhy is it not possible to predict whether H3PO4 or H2SO3 is the stronger acid? Show how you can use data to decide which is the stronger acid.arrow_forward
- Using Basicity to Determine If a Substitution Is Likely to Occur Will the following substitution reaction favor formation of the products?arrow_forwardWhat do you mean by carbon acid? What is the range of their pKa's?arrow_forwardRank each of the following sets of nitrogen bases in terms of basicity and explain your answerarrow_forward
- Arrange the compounds in each set in order of increasing base strength. consult Table 4.1 for pKa values of the conjugate acid of each base.arrow_forwardWhich proton in ketamine (C13H16ClNO) is the most acidic and what is the structure of its conjugate base?arrow_forwardWhich species is the stronger Lewis Acid? Why? (Use resonance structures to explain your answer.) BH3 or trimethoxyboratearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
