
Concept explainers
(a)
Interpretation:
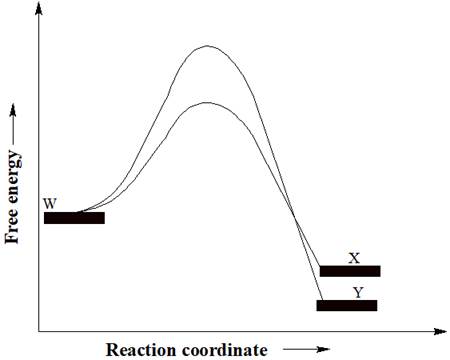
A reactant W can undergo two separate reactions to yield either X or Y. From the free energy diagram given below, the product that would be in greater abundance at equilibrium is to be determined.
Concept introduction:
In a reaction free energy diagram, Gibbs free energy is plotted as a function of the reaction coordinate. As the reaction coordinate increases, the geometries of the species involved in the reaction increasingly resemble those of the products. The product having lower energy signifies that it is the most stable product. The most stable product would be in greater abundance at equilibrium.
(b)
Interpretation:
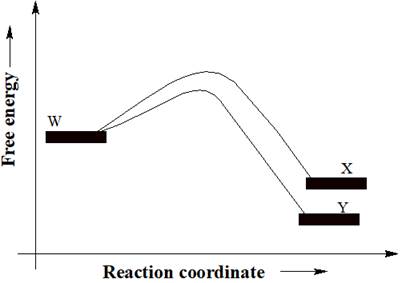
A reactant W can undergo two separate reactions to yield either X or Y. From the free energy diagram given below, the product that would be in greater abundance at equilibrium is to be determined.
Concept introduction:
In a reaction free energy diagram, Gibbs free energy is plotted as a function of the reaction coordinate. As the reaction coordinate increases, the geometries of the species, involved in the reaction, increasingly resemble those of the products. The product having lower energy signifies that it is the most stable product. The most stable product would be in greater abundance at equilibrium.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Select the reactions in Figure 39 that can be represented by the accompanying energy diagram?arrow_forwards sugar X a reducing or a non-reducing sugar? Reducing, because one/both of the rings can open to expose a free aldehyde group Non-reducing, because one/both of the rings can open to expose a free aldehyde group Reducing, because neither of the rings can open to expose a free aldehyde group Non-reducing, because neither of the rings can open to expose a free aldehyde group Reducing, because one/both of the rings can open to expose a free ketone group Non-reducing, because one/both of the rings can open to expose a free ketone group Reducing, because neither of the rings can open to expose a free ketone group Non-reducing, because neither of the rings can open to expose a free ketone group Cannot be determined from the structure shownarrow_forwardexplain electrophilic substitution reaction with example.arrow_forward
- Just to reiterate, the question is to draw out all possible alkene reactants that will result in the shown product.arrow_forwardDraw the curved arrow mechanism to explain the product formation for the following reactions, show how the reaction is catalytic and name all components.arrow_forwardUse curved arrow notation to draw mechanism, include stereochemistry, intermediates + final product and lone pairs.arrow_forward
- For each organic reaction, say whether at equilibrium there will be more reactants than products (so equilibrium lies ro the left), or more products than reactants (so equilibrium lies to the right)...arrow_forwardPredict the organic and inorganic products of the given reaction, and select the class to which the product belongs. Then, complete the curved‑arrow mechanism.arrow_forwardHow does this chair conformer make this other this in a synthesis reaction?arrow_forward
- Why does Hammett Equation only apply to meta and para substituted rings and not others? Explainarrow_forwardWhich of the reactions in has the most kinetically stable product?arrow_forwardWhat is the overall reaction called? Which step in the backward direction releases the most free energy?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
