
Concept explainers
To cool a summer home without using a vapor compression refrigeration cycle, air is routed through a plastic pipe
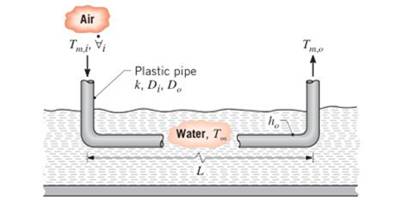
If air from the home enters the pipe at a temperature of
Trending nowThis is a popular solution!

Chapter 8 Solutions
Fundamentals of Heat and Mass Transfer
Additional Engineering Textbook Solutions
INTERNATIONAL EDITION---Engineering Mechanics: Statics, 14th edition (SI unit)
Mechanics of Materials (10th Edition)
Fundamentals Of Thermodynamics
Engineering Mechanics: Dynamics (14th Edition)
Applied Fluid Mechanics (7th Edition)
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
- A cuboid-shaped device has the following attachments: one entrance pipe and two exit pipes. Air enters the entrance pipe at 2.1 MPa, 355 degrees Celsius, and 126 m/s. In one of the exit pipes, the air leaves at 152 kPa, 160 degrees Celsius, and 45 m/s. In the other exit pipe, air leaves at 0.40 MPa and 220 degrees Celsius. The inside diameter of the pipes are as follows: Entrance pipe = 8 cmExit pipes (identical inside diameter) = 11 cmAssuming a steady-state, steady-flow condition, determine the mass flux (kg/s) and velocity (m/s) of air at the condition 0.40 MPa and 220 degrees Celsius exit pipe. Further, assume the air is an ideal gas.arrow_forwardDefine coefficient of performance (COP). The food compartment of a refrigerator as shown in figure is maintained at 4 °C by removing heat from it at a rate of 360 kJ/min. If the required power input to the refrigeration is 2 kW, determine the coefficient of performance of the refrigerator. Kitchen R QH QL= net,in = 2 kW = 360 kJ/min Food compartment 4°Carrow_forwardProblem 7: A power plant operates between temperatures TH = 86°F and T = 41°F at a depth of 2100 ft. About 13,300 gpm of water is pumped through a 40-in diameter pipe for cooling. The water experiences a temperature rise of 6°F with a thermal efficiency of 2.5%. Determine the amount of power generated. Consider the density of the water to be 64 Ibm/ft.arrow_forward
- An electric windshield defroster is used to remove 0.25-in of ice from a windshield. The properties of the ice are Tsat = 328F, uif = hif = 144 Btu/lbm, and v = 0.01602 ft3/lbm. Determine the electrical energy required per square foot of windshield surface area to melt this ice and remove it as liquid water at 328F. What is the minimum temperature at which the defroster may be operated? Assume that no heat is transferred from the defroster or ice to the surroundings.arrow_forward(b) Explain what is wrong with the system shown in Figure Q4(b).arrow_forwardSteam at T1 = 320°C and h1 = 60 W/m2·°C flows in a cast iron pipe (k = 80 W/m·°C). The inner and outer diameters are D1 = 5 cm and D2 = 5.5 cm, respectively. The insulation thickness is 3-cm-glass wool insulation with k= 0.05 W/m · °C. The temp. of the surroundings at T2 = 5°C and heat transfer coefficient of h2=18 W/m2·°C. Determine 1. Heat loss from the steam per unit length of the pipe. 2. Determine the temperature at the surfaces of the pipe and the insulation.arrow_forward
- Water is to be pumped from the bottom of a well 20 ft deep and 160ft above the ground with a specific heat of 1 BTU/lbm F is pumped using a 1-hp jet pump. Heat is lost from the whole system at the constant rate of 500 BTU/min. What is the temperature (℉) of the water as it enters the vessel if the flow rate of the water is 5 gal/min?assuming that the well water is at 35℉? kindly show all necessary calculationsarrow_forward6. 1 - J: Water is passed through a 3 cm internal diameter, 8 m long tube is heated from 15 °C to 75°C . The tube is equipped with an electric resistance heater, which provides uniform heating throughout the surface of the tube. The outer surface of the heater is well insulated, so that in steady operation all the heat generated in the heater is transferred to the water in the tube. If the system is to provide hot water at a rate of 8 L/min, determine the power rating of the resistance heater. Also, estimate the inner surface temperature of the pipe at the exit. L 3 p= 990.1kg/m? k = 0.637 W/m.°C v = u/p=0.602 x10-6 m²/s C, = 4180 J/kg.°C Pr = 3.91arrow_forwardA steam power plant is located in the northern part of the country. Steam in the plant flows steadily through a 0.17-m diameter pipeline from the steam generating unit (SGU) to the steam turbine. At the SGU end, the steam conditions are determined to be P = 4 MPa, t = 400°C, h = 3,247 kJ/kg, and specific volume = 0.181 m³/kg. At the steam turbine end, the conditions are found to be P = 3.5 MPa, t = 392°C, h = 3,178.3 kJ kg, and specific volume = 0.248 m³/kg. Heat loss from the pipeline is 7.86 kJ/kg. Determine the steam mass flow rate in kg/s.arrow_forward
- Steam at T∞ 1 = 320°C flows in a cast iron pipe (k = 80 W/m · °C) whose inner and outer diameters are D1 = 5 cm and D2 = 5.5 cm, respectively (Figure Q2b). The pipe is covered with 3-cm-thick glass wool insulation with k = 0.05 W/m · °C. Heat is lost to the surroundings at T2 = 5°C by natural convection and radiation, with a combined heat transfer coefficient of h2 = 18 W/m2 · °C. Taking the heat transfer coefficient inside the pipe to be h1= 60 W/m2 · °C, determine; i) the rate of heat loss from the steam per unit length of the pipe; and ii) the temperature drops across the pipe shell and the insulation.arrow_forwardRefrigerant vapor rejects 70 kW of heat as it passes through an air-cooled condenser. The condenser has an air-side area of 210 m2 and, based on this area, has an overall heat transfer coefficient of 0.037 kW/m2∙°C. Cooling air flows at a rate of 7.59 kg/s. If condensation of the refrigerant occurs at 55°C, what is the outlet temperature (in °C) of the air? Take the specific heat of air to be 1.02 kJ/kg∙°C. Round your answer to 2 decimal places.arrow_forwardm = 0.9000 kg of water is heated with P = 481 W electric heater for t = 39 min. During this time the temperature of the water rises from T₁ = 335 K to its boiling point and me = 176 g of water evaporates. How much of the electric energy used by the heater is transferred to the water during the heating? Please consider that produced energy can be obtained by the product of power and time, so E = P t. Please use for the kJ specific enthalpy of evaporation hf = 2260 kJ and heat capacity of water values p = 4.1900 kgK What is the Percentage of electric energy transferred to water? EP = 1 % Insert only 3 most significant digits of your answer without rounding. Quantity Symbol Total mass of water Mass of water that evaporates Power of electric heater Heating time Percentage of electric energy transferred to water Initial temperature of water Boiling point of water Specific enthalpy of vaporization for water • ÉASZ SÁN m me P t EP T₁ Tb he Specific heat capacity of liquid water Electric…arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





