
Batch processes are often used in chemical and pharmaceutical operations to achieve a desired chemicalcomposition for the final product. Related heat transferprocesses are typically transient, involving a liquid a fixed volume that may be heated from room temperature to a desired process temperature, or cooled fromthe process temperature to room temperature. Considera batch process for which a pharmaceutical (the coldfluid. c) ¡s poured into an insulated, highly agitated vessel (a stirred reactor) and heated by passing a hot fluid(h) through a submerged heat exchanger coil of thin-walled tubing and surface area
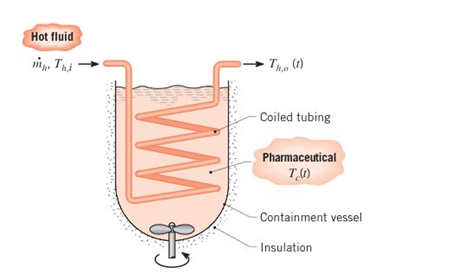
(a) Starting from basic principles, derive expressions thatcan be used to determine the variation of
(b) Consider a pharmaceutical of volume
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Fundamentals of Heat and Mass Transfer
Additional Engineering Textbook Solutions
Statics and Mechanics of Materials
DESIGN OF MACHINERY
Fundamentals Of Thermodynamics
Engineering Mechanics: Statics & Dynamics (14th Edition)
Vector Mechanics for Engineers: Statics
Thermodynamics: An Engineering Approach
- NUMBER 4 A food product wants to be produced in a small round shape (pellet) by freezing it in a water blast freezer freezer. Air freezer operates at -30 ° C. The initial product temperature is 25 ° C. The pellet has a diameter of 1.2 cm, and a density of 980 kg / m³. The initial freezing temperature is -2.5 ° C. The latent heat of freezing of the product is 280 kJ / kg. The thermal conductivity of the frozen product is 1.9 W / (m ° C). The convective heat transfer coefficient is 50 W / (m² K). Calculate the freeze time. t f = hourarrow_forwardhnen't For 1 mole of a gas, the van der Waals equation is where R is the gas constant (0.0821 L atm K mol ') and 7 is the Kelvin temperature 1The constants a and h are constants particular to a given gas and correct for the attractive forces between gas molecules, and for the volume occupied by the gas molecules, respectively. For methane (CH), the constants are a 2.253 L'atm and b4.278 x 10 L. Using the rearranged form of the van der Waals equation RT V -b v? calculate the pressure of 1 mole of methane as a function of container volume 0°C (273 K) at suitable volumes from 22.4 L to 0.05 L. Use one of the at custom functions described in this chapter to calculate the first and second derivatives of the P-V relationship. Compare with the exact expressions dP RT 2a dV (v - b)?v3 d? P 2RT 6a dv? (V -b)arrow_forward17 lı. 9:08 A docs.google.com 5ialy čhāi For producing 1 ton of crude (pig) iron in blast furnace, the quantity of iron * :ore is 0.65 ton 0.2 ton O 0.05 ton O 1.6 ton O ialy ihäi Refrigerator liners, appliance housings, * :and shower stalls are producing by Injection Molding O Extrusion process O Rotational Molding O Thermoforming O iialy ibai The capacity of a . was from .8 to 30 tons of molten iron Oxygen Convertor Open Hearth O Bessemer Convertor O Electrical furnace O نقطة واحدة The polymer production process which including chemical reaction between two polymer materials - * :thermoset is Rotational Molding O Thermoforming Blow Molding O Reaction Injection Molding O O O O O O Oarrow_forward
- The TPD method measures temperature elevations in a tissue region during a heating pulse and its later temperature decay after the pulse. It is then using the Pennes bioheat equation to perform a curve fitting to determine the local blood perfusion rate. If the TPD probe is placed in the vicinity of very large blood vessel, will the TPD technique provide an accurate measurement of the local blood perfusion in the vicinity of this large blood vessel? Explain briefly. (Hint: Is the Pennes bioheat equation accurate surrounding a large blood vessel?)arrow_forwardQu: Ball bearing ro-4 mm leaving the oven at a uniform temperature of 900 °C ar exposed to air for a while before they are dropped in to the water for quenching Determine the time they can stand the air before their temperature falls below 850 also find the total rate of heat transfer .Take for the balls k 54 W/m .°C , p= 783 kg/m', C,-0.456 kJ/kg.°C %3D Ans : 163 s , 543 W Furmacearrow_forwardSubject : Heat Transfer Solve the surface temperature using the parameters: h steam = 17.6319 W/m2 Kh air = 25 W/m2 KT steam = 120 deg CT air = 30 deg C k of cooling fill= 0.147 W/(m.K) Area of fill = 128.64 m'2 thickness of cooling = 2marrow_forward
- 6 In a boiler test, the following observations wcre made: 358 A Text Book of Thermal Engineering Feed water temperaturç e= 12" C; Pressure of steam = 11 bar; Dryness fraction of steam 095 Mass of coal burnt 300 kg/h, Calorific value of coal 32 000 kJkg of coal; Mass of water supplied to boiler in 7 hrs 14 min= 14 625 kg. The mass of w ster in the boiler at the end of the test was less than that at the commencement by 900kg. Calculate I. Actual evaporation per kg of coal: 2 Equivalent evaporation from and at 100 C per kg [Ans. 7 15 kg, 8 33 kg: 58.75 % of coal ; and 3. Thermal efficiency of the boilerarrow_forwardA cylindrical liquid oxygen (LOX) tank has a diameter of 1.22 m, a length of 6.1 m, and hemispherical ends. The boiling point of LOX is -179.4C. An insulation is sought that will reduce the boil-off rate in the steady state to no more than 11.3 kg/h. The heat of vaporization of LOX is 214 kJ/kg. If the thickness of this insulation is to be no more than 7.5 cm, what would the value of its thermal conductivity have to be?arrow_forwardRecall that a there fore thermodynamie detinihon Constant pressure, AH = 9. We can from the caleulate AS for any process S= arev こ ナ Use this informmaton to deprive dAS an expressiom for but temperature - Invariant heat capacuty change s Cp. Integrate this equathon Such that Calculate the AS at any temp T given a known value AS, at To for a process Involving a non-zero you Canarrow_forward
- A liquid reactant solution is pumped through a pre-heater before entering a reaction vessel. The solution enters the pre-heater at 25 °C and must enter the reactor at 45 °C. The flow of the reactant solution is 1500 mL per minute. The density of the reactant solution is 0.91 g-cm3 and the heat capacity is 1.75 Jg1.K1. ASsume there is no heat loss in the pre-heater or between the pre-heater and the reactor. a. Draw a schematic of the pre-heater, detailing all mass and energy flows. b. Clearly define a system boundary. c. Write the general energy balance, and a simplified energy balance stating all assumptions that apply to this problem. d. What is the minimum power rating required for the pre-heater?arrow_forwardPROBLEM #5 gaa is being air film at & total 'pressure of 1:l bar- The paitial att 'one boundang at the other, The partial pressure remains com tant a transfered across a staghant pressure of the gas is' 0.45 bar of the film and 0. 12 bar a Find: an) effect m diffusiom coefficient if mass transfer rate'doubles bi) total pressure mass transter rate heeded to dowble the of the gasarrow_forwardWrite down the one dimensional heat flow equation in unsteady state.arrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
