
Concept explainers
(a)
Interpretation:
The hemiacetal in glucosamine should be labeled.
Concept Introduction:
The addition of one molecule of alcohol to an
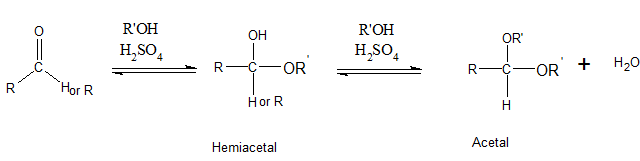
(b)
Interpretation:
All the functional groups in glucosamine should be labeled.
Concept Introduction:
The addition of one molecule of alcohol to an aldehyde or ketone results in a hemiacetal. Here, the C=O double bond breaks and single bonds are formed. If the hemiacetal is acyclic, it is not stable. Acyclic hemiacetal reacts with a second alcohol molecule and forms acetal.
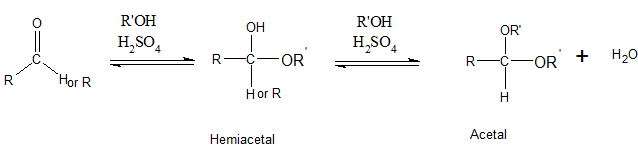
Even though acetals contain C-O-R bond, acetals are not ethers. Acetals have 2 OR groups attached to a single C atom. But an ether has one O atom which is bonded to 2 carbons.
Alcohols are compounds which have OH groups.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Why is thymol insoluble with bicarbonate and diethyl ether and why is glycine soluble in HCl? Thank you in advance!arrow_forwardCholic acid, a major constituent of bile, has the structure shown.(a) Draw the structure of cholic acid, showing the rings in their chairconformations, and label each methyl group and hydroxy group asaxial or equatorial. (Making a model may be helpful.)(b) Cholic acid is secreted in bile as an amide linked to the aminogroup of glycine. This cholic acid–amino acid combination acts asan emulsifying agent to disperse lipids in the intestines for easierdigestion. Draw the structure of the cholic acid–glycine combination,and explain why it is a good emulsifying agent.arrow_forwardWhy are acetic acid, sodium acetate, and sodium caprate all soluble in water, whereas capric acid, a 10-carbon fatty acid, is not?arrow_forward
- (a) Locate the glycosidic linkage in cellobiose. (b) Number the carbon atoms in both rings. (c) Classify the glycosidic linkage as α or β, and use numbers to designate its location.arrow_forwardGive the detailed pathway for the biosynthesis of melatonin, including the type of reaction and the catalysts involved in each step and clear chemical structure.arrow_forwardHow do you know which is the R and which is the S ibuprofen?arrow_forward
- Explain about Stemoamide ?arrow_forward(i) Acetylation of glucose with acetic anhydride gives glucose pentacetate. Write the structure of the pentacetate. (ii) Explain – Why glucose pentacetate does not react with hydroxylamine?arrow_forwarda. Which box contains a carbocyclic unsaturation? b. Which box contains an aryl structure? c. Which box contains carboxyl group? d. Which box contains a sulfide group? e. Which box contains an amide group?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
