
Concept explainers
(a)
Interpretation:
The structure of the salmeterol should be drawn.
Concept Introduction:
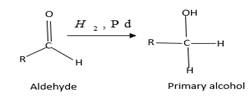
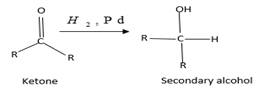
(b)
Interpretation:
One chirality center in a salmeterol molecule should be identified.
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
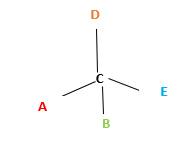
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different functional groups.
(c)
Interpretation:
Enantiomers of salmeterol in three dimensions should be drawn.
Concept Introduction:
Mirror image of a molecule is a reflected duplicate of the molecule.
Enantiomers − the mirror image of the original molecule of a chiral molecule. These are stereoisomers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- The structure of 4 isomers of ketopentose are shown. 1) select every structure that is a diastereomer of structure D A, B, or C? 2) select every structure that is a enantiomer of structure C A, B, or D 3) select every structure that is a stereoisomer of structure C A, B, or Darrow_forwardWhat are diastereomers? a. Constitutional isomers. b. Molecules with only one chiral center. c. Stereoisomers that are not mirror images of one another and are non-superimposable on one another. d. Stereoisomers that are mirror images of one another.arrow_forwardCaptopril is a drug used to treat high blood pressure and congestiveheart failure.Draw the enantiomer of captoprilarrow_forward
- A(n) ________ is an achiral compound that contains chiral centers but is superimposable on its mirror image. A) constitutional isomers B) conformational isomers C) enantiomers D) diastereomers E) meso compoundsarrow_forwardOne of the analgesics has a chiral center. Which compound is it? One of the two enantiomers is far more effective at reducing pain than the other.arrow_forwardCelery ketone, like carvone, has two distinct aromas. In contrast to the S enantiomer's licorice scent, the R enantiomer has an earthy, celery-like aroma. Each enantiomer should be depicted and its odor assigned.arrow_forward
- The structure of 4 isomers of an aldotetrose carbohydrate are given. 1) select every structure that is a diastereomer of structure D A, B, or C? 2) select every structure that is a enantiomer of structure C D, B, or A? 3) select every structure that is a stereoisomer of structure D A, B, or Carrow_forwardRe-draw each Fischer projection formula using wedges and dashed wedgesfor the stereogenic center, and label the center as R or S.arrow_forward(a) assign R or S configuration to each chiral center, (b) Which compound are enantiomers? (c) Which compounds are diastereomers?arrow_forward
- Captopril is a drug used to treat high blood pressure and congestive heart failure. a.Designate each stereogenic center as R or S. b.Draw the enantiomer of captopril. c.What product is formed when captopril is treated with one equivalent of NaH? d.What product is formed when captopril is treated with two equivalents of NaH?arrow_forwardA. Assign each chiral center as R or S B. draw a diastereomer of ephedrine.arrow_forwardDraw the structure of (S)-citalopram, a drug used to treat depression and anxiety that is much more potent than its R enantiomer.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




