
Concept explainers
(a)
Interpretation:
The alcohol formed due to reaction of carbonyl with
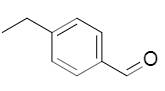
Concept introduction:
In the reduction of carbonyl compounds, hydrogen atoms are added to the carbonyl carbon, forming alcohols.
(b)
Interpretation:
The needed structural differences should be made to 4-methylcyclopentanone to derive the relevant product of reduction.
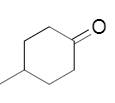
Concept introduction:
In the reduction of carbonyl compounds, Hydrogen atoms are added to the carbonyl carbon, forming alcohols finally. Ketones form secondary alcohols.
(c)
Interpretation:
The needed structural differences should be made to 5-methyl-3-hexanone to derive the relevant product of reduction.
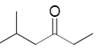
Concept introduction:
In the reduction of carbonyl compounds, ketones form secondary alcohols.
(d)
Interpretation:
The needed structural differences should be made to Octanal to derive the relevant product of reduction.
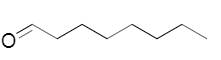
Concept introduction:
In the reduction of carbonyl compounds, an aldehyde is converted to primary alcohol.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What alcohol is formed when each compound is treated with H 2 and a Pd catalyst?arrow_forwardWhat alcohol is formed when the compound is treated with H2 and a Pd catalyst?arrow_forwardn-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forward
- Give the structure of an alcohol that could be used to prepare each of the following compounds: a. b. c.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardDraw the structures of the ethers that can be produced from the following alcohols: a. CH3CH2CH2OH b. c.arrow_forward
- Draw the structures of the chief product formed when the following alcohols are dehydrated to alkenes: a. b.arrow_forwardA primary alcohol has a hydroxyl group bonded to a(n) a. aromatic carbon. b. trisubstituted carbon. c. disubstituted carbon. d. singly substituted or unsubstituted carbon.arrow_forwardWhat kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholsarrow_forward
- Glycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forwardWhy are lower molecular weight alcohols more soluble in water than higher molecular weight alcohols?arrow_forwardWhat alcohol is formed when attached compound is treated with NaBH4 in CH3OH?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


