
Concept explainers
(a)
Interpretation:
The carbonyl compound needed to produce the following alcohol by a reduction reaction should be determined:
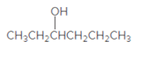
Concept Introduction:
In a reduction of an organic molecule, either the number of C-O bonds decreases or number of C-H bonds increases. So when a
(b)
Interpretation:
The carbonyl compound needed to produce the following alcohol by a reduction reaction should be determined:
(CH3)2CHCH2CH2OH
Concept Introduction:
In a reduction of an organic molecule, either the number of C-O bonds decreases or number of C-H bonds increases. So when an
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Give the structure corresponding to each IUPAC name. 2,4 dimethyl- 2 hexanolarrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardALCOHOLS 1. WHY IS ETHANOL MORE SOLUBLE IN WATER THAN 1-HEXANOL? 2. WHAT IS DENATURED ALCOHOL? AND WHY IS ALCOHOL DENATURED? ETHER 1. WHY DOES DIETHYL ETHER HAVE MUCH LOWER BOILING POINT THAN 1-BUTANOL?arrow_forward
- What alcohol is formed when each compound is treated with NaBH4 in CH3OH?arrow_forwardn-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forwardExplain Addition of Alcohols—Acetal Formation ?arrow_forward
- What carbonyl compound is needed to make each alcohol by a reduction reaction? What carbonyl compound is needed to make each alcohol by a reduction reaction?arrow_forwardRank the alcohols in order of increasing reactivity when dehydrated with H2SO4.arrow_forwardWhat alkenes are formed when attached alcohol is dehydrated with TsOH?Label the major product when a mixture resultsarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

