
Concept explainers
(a)
Interpretation:
The skeletal structure of the given compound should be drawn.
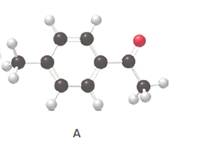
Concept introduction:
A carbonyl compound in the given diagram is represented as a stick ball model. In stick ball model, atoms are represented as balls and bonds are represented as sticks.
(b)
Interpretation:
An acceptable name for A should be determined.
Concept introduction:
The given compound can be named using rules for IUPAC naming.
(c)
Interpretation:
The number of trigonal planar carbon in the given compound should be determined.
Concept introduction:
Trigonal planar carbon atoms possess sp2 hybridization.
(d)
Interpretation:
The products formed when A is reacted with two equivalent molecules of CH3OH in the presence of acid should be determined.
Concept introduction:
When an
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 1. What are the characteristics of a positive tollens test for adehydes? What is the oxidizing agent in tollens solutions? 2. What is the characteristics of a positive Benedict's test for aldehydes? What is the oxidizing agent in Benedict's solution?arrow_forwardDraw the products formed when (CH3)2C=CH2 is treated with following reagent. Cl2arrow_forwardALCOHOLS 1. WHY IS ETHANOL MORE SOLUBLE IN WATER THAN 1-HEXANOL? 2. WHAT IS DENATURED ALCOHOL? AND WHY IS ALCOHOL DENATURED? ETHER 1. WHY DOES DIETHYL ETHER HAVE MUCH LOWER BOILING POINT THAN 1-BUTANOL?arrow_forward
- 6. Draw the correct structures for the following: a. what is the correct structure of 3-methyl 1-pentyne? b. what is the correct structure of butyl methyl amine? c. what is the correct structure of 3-methyl 1-pentyne? d. what is the correct structure of 2-methyl 2-butanol? e. what is the correct structure of pentanoic acid?arrow_forwardMaltose is a carbohydrate present in malt, the liquid obtained from barley and other grains. Although maltose has numerous functional groups, its reactions are explained by the same principles we have already encountered.a. Label the acetal and hemiacetal carbons.b. What products are formed when maltose is treated with each of these reagents: [1] H3O+; [2] CH3OH and HCl; [3] excess NaH, then excess CH3I?c. Draw the products formed when the compound formed in Reaction [3] of part (b) is treated with aqueous acid.The reactions in parts (b) and (c) are used to determine structural features of carbohydrates like maltose.arrow_forwardDraw the missing starting material. Reagent 1 is benzene and AlCl3. Reagent B is Zn(Hg) and HCl.arrow_forward
- #20 B Draw structural formulas for all possible carbocations formed by the reaction of each alkene with HCl.arrow_forwardn-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. H2SO4arrow_forward
- Draw the product formed when cyclohexane carbaldehyde is reacted with NaOH and H2O.arrow_forwardWhat product is formed when each alkene is treated with HCl?arrow_forward1.) What is the structure and IUPAC name of tert-butyl isopropyl acetylene? 2.) What is the structure of 2 methyl- dimethyl-alpha-hexylene? 2.) What is the structure of 3 methyl- dimethyl-beta-hexylene?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





