
Concept explainers
(a)
Interpretation:
The structure of the functional group in alkene family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes
Alkenes are one of the important types of hydrocarbon which have at least one carbon-carbon double bond (
(b)
Interpretation:
The structure of the functional group in alcohol family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes alcohol, aldehyde, amine, ether, carboxylic acid etc.
Alcohol: It is an organic compound where it contains at least one
(c)
Interpretation:
The structure of the functional group in ester family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes alcohol, aldehyde, amine, ether, carboxylic acid etc.
Ester: Ester is a compound which is derived from an acid in which at least one hydroxyl group is replaced by a
(d)
Interpretation:
The structure of the functional group in amine family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes alcohol, aldehyde, amine, ether, carboxylic acid etc.
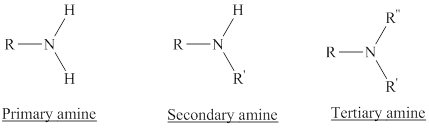
Amines: They are the derivatives of ammonia with one or more of the hydrogens replaced with a substituent such as an alkyl group.
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
General Chemistry: Atoms First
- which functional groups are in C6H12O2 with it having 1 degree of saturation, and is the molecule hexanoic acid?arrow_forwardGlucose, C6H12O6, contains an aldehyde group but exist predominantly in the form of the cyclic hemiacetal show below. A cyclic hemiacetal is formed when the —OH group of one carbon bonds to the carbonyl group of another carbon. Identify which carbon provides the —OH group and which provides the —CHO? Give a functional isomer of glucose and draw its structure.arrow_forwardThere are eight structures with the formula C3H7NO in whichthe O is part of a carbonyl group. Draw the structures andidentify the functional groups in each.arrow_forward
- Give the molecular formula of a hydrocarbon containingsix carbon atoms that is (a) a cyclic alkane, (b) a cyclicalkene, (c) a linear alkyne, (d) an aromatic hydrocarbon.arrow_forwardDraw the condensed structures of the compounds formedfrom (a) butanoic acid and methanol, (b) benzoic acid and2-propanol, (c) propanoic acid and dimethylamine. Namethe compound in each case.arrow_forwardDescribe the structure of the carboxyl group. Complete the following table:arrow_forward
- What is the full structure of an amine which is an isomer of ethylamine? What is the isomer called? Is it primary, secondary or tertiary?arrow_forwardGive the molecular formula of a hydrocarbon containing six carbon atoms that is a cyclic alkane.arrow_forwardHow do you distinguish between geometrical and structural isomers? Give examples.arrow_forward
- What structural features help us identify a compound as(a) an alkane, (b) a cycloalkane, (c) an alkene, (d) an alkyne,(e) a saturated hydrocarbon, (f) an aromatic hydrocarbon?arrow_forwardWrite the structural formula of methylcyclopentenearrow_forward(a) What is meant by the term isomer ? (b) Among the four alkanes, ethane, propane, butane, and pentane, which is capable of existing in isomeric forms?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
