
Concept explainers
Interpretation:
The functional groups present in novocaine have to be identified. Also the structure of carboxylic acid and alcohol that are needed to form novocaine has to be drawn.
Concept Introduction:
Carboxylic acid: One
Ester: One
Esterification reaction: Esters are prepared by the reaction of a carboxylic acid and an alcohol molecule with the elimination of water molecule. The addition of a strong acid such as
Here, the
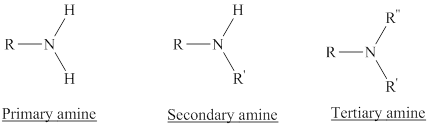
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Aromatic Compounds: Compounds that are planar, conjugate, cyclic and having
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
General Chemistry: Atoms First
- The functional group —COOH is found in a. esters b. aldehydes c. alcohols d. organic acidsarrow_forwardexplain the structures and properties of functional group :carboxylic acid and aminoarrow_forwardWhat are fats and oils? a. ketones formed of two or more esters b. any large molecular weight organic compound that has been saturated c. esters formed from glycerol and three carboxylic acids d. polymers of repeating aromatic hydrocarbonsarrow_forward
- Given the structure of Novocain, drawn the structures of the alcohol and carboxylic acid that would react to produce novocain.arrow_forwarda. What type(s) of intermolecular forces are expected between benzyldiethylamine molecules?arrow_forwardWhat functional group is present in the reactants (isobutanol and propionic acid) but not the ester (isobutyl propionate) ?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





