
Concept explainers
(a)
Interpretation: To identify whether the statement “four of the 20 standard amino acids contain sulfur” concerning sulfur-containing amino acids is true or false.
Concept introduction: Amino acids are the
There are 20 amino acids present in nature. These are arginine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, valine, alanine, asparagines, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, and tyrosine.
(b)
Interpretation: To identify whether the statement “the first step in the degradation of the amino acid cysteine is a transamination reaction” concerning sulfur-containing amino acids is true or false.
Concept introduction: There are 20 amino acids present in nature. These are arginine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, valine, alanine, asparagines, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, and tyrosine. Only two of the twenty amino acids have a sulfur atom in their structure. The two amino acids are cysteine and methionine. The structure of cysteine is:
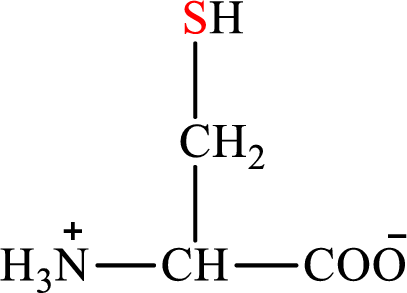
Cysteine gives pyruvate as the degradation product. The degradation of cysteine is a two-step process.
(c)
Interpretation: To identify whether the statement “pyruvate is the sulfur-containing product from the degradation of the amino acid cysteine” concerning sulfur-containing amino acids is true or false.
Concept introduction: There are 20 amino acids present in nature. These are arginine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, valine, alanine, asparagines, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, and tyrosine. Only two of the twenty amino acids have a sulfur atom in their structure. The two amino acids are cysteine and methionine. The structure of cysteine is:
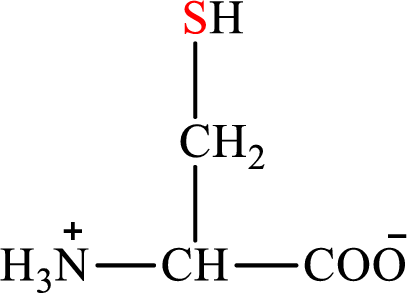
Cysteine gives pyruvate as the degradation product. The degradation of cysteine is a two-step process.
(d)
Interpretation: To identify whether the statement “ATP is the activating agent in the first step of the conversion of serine to cysteine” concerning sulfur-containing amino acids is true or false.
Concept introduction: There are 20 amino acids present in nature. Only two of the twenty amino acids have a sulfur atom in their structure. Cysteine is biosynthesized from nonessential amino acid serine. The structure of cysteine is:
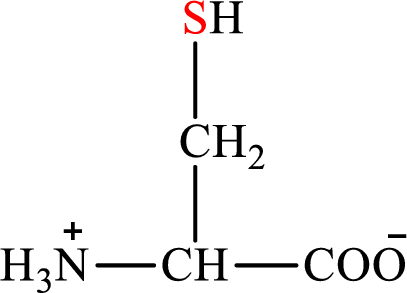
The structure of serine is:
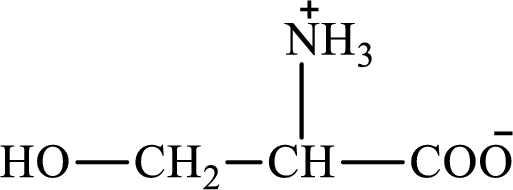
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Which of the following statements concerning the enzyme regulation is CORRECT? Select one: A. Citrate is the allosteric inhibitor of hexokinase. B. AMP is the alloseric activator of pyruvate dehydrogenase. C. Glucose-6-phosphate is the allosteric activator for phosphofructokinase. D. ATP is the activator of pyruvate kinase.arrow_forwardWhich of the following statements about the "central role" of TCA is/are correct? a. Some metabolic intermediates of TCA are used as non-carbohydrate sources for gluconeogenesis. b. β-oxidation of lipids produces acetyl-CoA that can enter TCA. c. All metabolic intermediates in TCA are used in the biosynthesis of amino acids. d. Degraded amino acids are converted to metabolic intermediates of TCA, then to ketone bodies.arrow_forwardWhich of the following statements is true of the β - oxidation of an unsaturated fatty acid? a. It is identical to the β-oxidation of a saturated fatty acid. b. It involves an extra step to convert a cis double bond to a trans double bond. c. It involves an extra step to convert a trans double bond to a cis double bond. d. It is completely different from the β-oxidation of a saturated fatty acid.arrow_forward
- Which of the following is NOT involved in the activation of glucose for glycogen synthesis? A. Phosphorylation of glucose by hexokinase to glucose-6-phosphate. B. Isomerization of glucose-6-phosphate to fructose-6-phosphate by phosphohexose isomerase. C. Two-step phosphoryl group transfer by phosphoglucomutase. D. Attachment of glucose-1-phosphate to UDP.arrow_forwardOrder the steps that results in a triacylglycerol containing two palmitic acids and one stearic acid. a. Diacylglycerol reacts with an fatty-acyl-CoA b. Phosphate is remove from phosphatidic acid to form diacylglycerol c. Two of the fatty acyl-CoAs react with glycerol-3-phosphate d. An elongase produce stearic acid e. Stearoyl-CoA is synthesized…arrow_forwardWhich of the following produces the highest number of reducing equivalent molecules per mole of a saturated 10 carbon fatty acid chain in aerobic conditions? A. Oxidation of Malate to Oxaloacetate B. the Electron Transport Chain C. Oxidation of a-Ketoglutarate to Succinyl-COA D. the B-Oxidation "spiral"arrow_forward
- The rate-limiting step in fatty acid synthesis is: a. The reduction of the acetoacetyl group to a ?-hydroxybutyryl group b. Formation of malonyl-CoA from malonate and coenzyme A. c. Condensation of acetyl-CoA and malonyl-CoA. d. The reaction catalyzed by acetyl-CoA carboxylase.arrow_forwardDefine the following terms: a. ketogenic amino acid b. glucogenic amino acid c. L-amino acid oxidase d. Krebs urea cycle e. CPSIarrow_forwardDifferentiate fatty acid synthesis and degradation in terms of the following: Fatty acid synthesis Fatty acid degradation a. Subcellular location b. End product c. Co-factors d. Carriers of acyl/acetyl groups between the mitochondria and the cytosolarrow_forward
- Indicate which of the following amino acids are ketogenic and which are glucogenic: a. tyrosine b. lysine c. glycine d. alanine e. valine f. threoninearrow_forward1. Explain the reaction mechanism involved how glucogenic amino acids can yield either a pyruvic acid or an oxaloacetic acid. In what pathway will pyruvic or oxaloacetic acid be used and why is this pathway important? 2. Discuss the reaction mechanism involved how the -NH2 groups of amino acids are being metabolized. 3. Explain why gluconeogenesis under conditions of starvation or diabetes breaks down body proteins. Complete answer please. Thank you. |arrow_forwardWhich of the following statements describing the mobilization and utilization of fatty acids in the body is INCORRECT? Answers A-E A. Fatty acids are excellent fuels for skeletal muscle. B. On a molar basis, fatty acid oxidation to carbon dioxide and water yields more ATP than do carbohydrates C. It is Coenzyme A derivatives of fatty acids that are beta-oxidized D. Fatty acids are transported in plasma bound to albumin E. Fatty acid oxidation to carbon dioxide and water does not require a functional Krebs cyclearrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





