
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 6IQ
- a. Draw the amino acids alanine (R group: −CH3) and serine (R group: −CH2OH) and then show how a dehydration reaction will form a peptide bond between them.
- b. Which of these amino acids has a polar R group? a nonpolar R group?
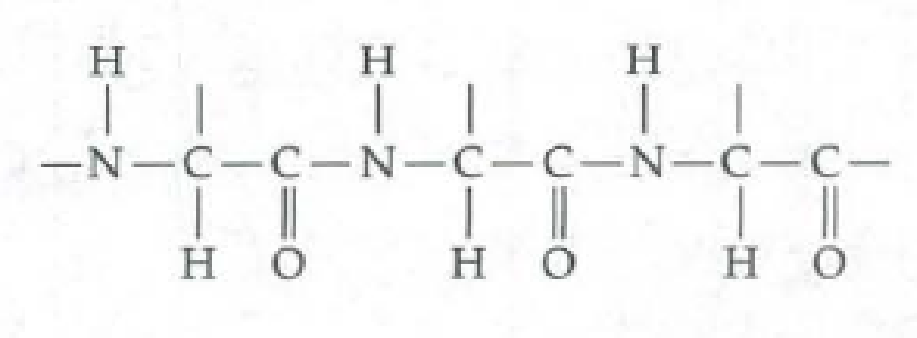
- c. What does the following molecule segment represent? (Note the N—C—C—N—C—C sequence.) What would be attached at each of the three vertical bond lines?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Refer to the figure below.
Replacing lysine with another amino acid in the protein may alter the shape and function of the protein. Replacing lysine with which type(s) of amino acid(s) would lead to the least amount of change in the tertiary structure of this protein? Explain.
Our growing understanding of how proteins fold allows researchers to make predictions about protein structure based on primary amino acid sequence data. Consider the following amino acid sequence.(a) Where might bends or β turns occur?(b) Where might intrachain disulfide cross-linkages be formed?(c) Assuming that this sequence is part of a larger globular protein, indicate the probable location (external surface or interior of the protein) of the following amino acid residues: Asp, Ile, Thr, Ala, Gln, Lys. Explain your reasoning.
(a) How is ubiquitination attached to the protein of interest? At which amino acid residue(s)? What enzyme(s) is involved, if any?(b) Is the ubiquitination relatively stable or highly dynamic? how does ubiquitination become detached from the protein of interest? what enzyme is involved?(c) what is the function of ubiquitination
Chapter 5 Solutions
Study Guide for Campbell Biology
Ch. 5 - Monomers are linked into polymers by ________...Ch. 5 - You can recognize a monosaccharide by its multiple...Ch. 5 - Number the carbons in the following glucose and...Ch. 5 - Prob. 4IQCh. 5 - Fill in this concept map to help you organize your...Ch. 5 - a. Draw the amino acids alanine (R group: CH3) and...Ch. 5 - In the following diagram of a portion of a...Ch. 5 - Now that you have gained experience with concept...Ch. 5 - a. Label the three parts of this nucleotide....Ch. 5 - Take the time to create a concept map that...
Ch. 5 - Prob. 1SYKCh. 5 - Prob. 2SYKCh. 5 - glycogen A. carbohydrate B. lipid C. protein D....Ch. 5 - cholesterol A. carbohydrate B. lipid C. protein D....Ch. 5 - RNA A. carbohydrate B. lipid C. protein D. nucleic...Ch. 5 - collagen A. carbohydrate B. lipid C. protein D....Ch. 5 - hemoglobin A. carbohydrate B. lipid C. protein D....Ch. 5 - A. carbohydrate B. lipid C. protein D. nucleic...Ch. 5 - Prob. 7TYKMCh. 5 - enzyme A. carbohydrate B. lipid C. protein D....Ch. 5 - cellulose A. carbohydrate B. lipid C. protein D....Ch. 5 - Chitin A. carbohydrate B. lipid C. protein D....Ch. 5 - Polymerization (the formation of polymers) is a...Ch. 5 - Which of the following statements is not true of a...Ch. 5 - Prob. 3TYKCh. 5 - Prob. 4TYKCh. 5 - Prob. 5TYKCh. 5 - Prob. 6TYKCh. 5 - A fatty acid that has the formula C16H32O2 is a....Ch. 5 - Prob. 8TYKCh. 5 - Prob. 9TYKCh. 5 - Prob. 10TYKCh. 5 - Which of the following molecules provides the most...Ch. 5 - Prob. 12TYKCh. 5 - What happens when a protein denatures? a. Its...Ch. 5 - The helix of proteins is a. part of a proteins...Ch. 5 - What is the best description of the following...Ch. 5 - Prob. 16TYKCh. 5 - Prob. 17TYKCh. 5 - Which of the following is true of the subunits of...Ch. 5 - Prob. 19TYKCh. 5 - If the nucleotide sequence of one strand of a DNA...Ch. 5 - How are nucleotide monomers connected to form a...Ch. 5 - Prob. 22TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- 1.a.If this molecule was the side chain of an amino acid, in a protein, what tertiary structure stabilizers could be present? b.Make a key and use color to highlight what part of you structure can be stabilized in each manner?arrow_forwardConsider the following peptide to answer the questions below: A M A K K E S H I F T A I D E What is the total charge of this peptide at pH = 5? What is the N terminal amino acid (full name)? C terminal amino acid? How many fragments would result if this peptide underwent proteolytic cleavage with V-8 protease and what are these fragments (list them)? Would this peptide be a candidate to form an alpha helix? Why or why notarrow_forwardDraw the polypeptide GRADE, write its full name and net charge at pH 7. How many peptide bonds are present, and which amino acid is on the C-terminus?arrow_forward
- 4. Amino acids are linked together by peptide bonds to form polypeptides. (a) Draw the structure of a tripeptide (b) draw an asterisks (*) directly beside all alpha carbons and draw a square around all peptide bonds (c) Use arrows to identify and label the N-terminus and C-terminus (d) Draw a circle around the R group of each amino acid residue in the tripeptide and classify each R group as polar, non polar, acidic, or basic (e) Using the abbreviated names of the amino acids, provide the primary structure of the tripeptide you have drawn.arrow_forwardGiven this short polypeptide: Met-Ala-Gly-Ser A) Which is the amino terminal amino acid? B) How many amino acids are in this polypeptide? C) How many peptide bonds are in this polypeptide? D) Which amino acid has the smallest side chain? E) Which amino acid contains sulfur?arrow_forwardWhich of the following characterize -helix regions of proteins? (A) They all have the same primary structure. (B) They are formed principally by hydrogen bonds between a carbonyl oxygen atom in one peptide bond and the amide hydrogen from a different peptide bond. (C) They are formed principally by hydrogen bonds between a carbonyl atom in one peptide bond and the hydrogen atoms on the side chain of another amino acid. (D) They are formed by hydrogen bonding between two adjacent amino acids in the primary sequence. (E) They require a high content of proline and glycinearrow_forward
- Valine is an amino acid with a nonpolar side chain and serine is one with a polar side chain. Draw the two amino acids.(a) Why is the side chain for valine nonpolar, whereas the side chain for serineis polar?(b) Which amino acid has a hydrophilic side chain and which has a hydrophobic sidechain?arrow_forwardRefer to the figure below. Replacing lysine with another amino acid in the protein may alter the shape and function of the protein. Replacing lysine with which type(s) of amino acid(s) would lead to the least amount of change in the tertiary structure of this protein? Explain your answer.arrow_forwardA. Where would you expect valine, leucine, and isoleucine to be located in a polypeptide? (Inside or outside)? Explain your answer using the properties of the amino acids listed. B.How would your answer change, if the amino acids were Lysine and Histidine?arrow_forward
- Which of the following is true A) The disulfide bridges formed by reduction of the sulfhydryl groups on cysteine destabilizes protein tertiary structure. B) The disulfide bridges formed by oxidation of the sulfhydryl groups on cysteine destabilizes protein tertiary structure. C) The disulfide bridges formed by oxidation of the sulfhydryl groups on cysteine stabilizes protein tertiary structure. D) The disulfide bridges formed by reduction of the sulfhydryl groups on cysteine stabilizes protein tertiary structure.arrow_forwardGive typing answer with explanation and conclusion On paper draw a dipeptide, clearly showing the peptide bond joining the two amino acids together. If the two amino acids are valine and threonine, predict the overall charge of the dipeptide at pH 7. Do not forget to consider the amino (N-terminal) and carboxy (C-terminal) of the dipeptide, as well as the R groups. Select one: a. +2 b. -2 c. 0 d. -1 e. +1arrow_forwardDraw the structural formula of the oligopeptide if the amino acids are arginine, glutamine, glycine, methionine and glutamic acid considering the first amino acid is the N-terminusarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY