
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 3IQ
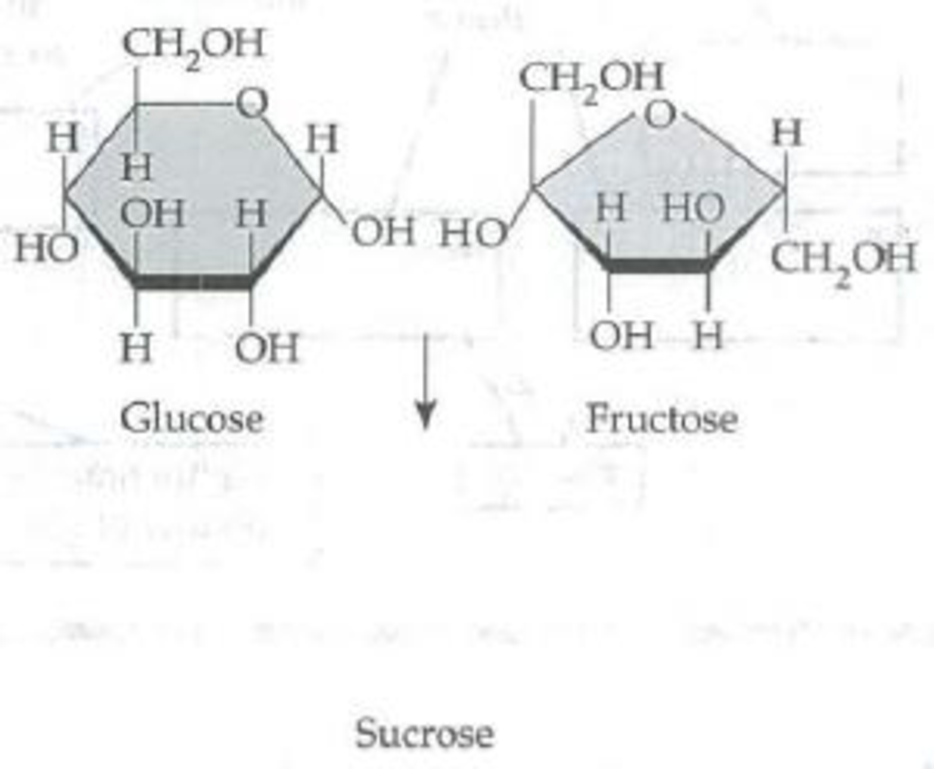
Number the carbons in the following glucose and fructose molecules (each unlabeled corner of the ring represents a carbon. In glucose, carbon 1 is to the right of the O in the ring; in fructose, carbon 1 extends up from the ring on the left side.) Circle the atoms that will be removed by a dehydration reaction. Then draw the resulting sucrose molecule with its 1–2 glycosidic linkage.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following describes lactose?
A. A nonreducing disaccharide containing glucose and fructose linked by a (α1-->4) bond.
B. A reducing disaccharide containing two glucose molecules linked by a (α1-->4) bond.
C. A reducing disaccharide containing two glucose molecules linked by a (β1-->4) bond.
D. A reducing disaccharide containing galactose and glucose linked by a (β1-->4) bond.
E. A nonreducing disaccharide containing galactose and glucose linked by a (α1-->4) bond.
a. Is the disaccharide below a non-reducing sugar? yes or no
b. The glycosidic linkage in the disaccharide is what β-(1-2) linkage, α-(1-2) linkage, β-(1-4) linkage, or α-(1-4) linkage?
c. The products of hydrolysis of the disaccharide (which of the ff.)
α-galactose and α-ribose
α-glucose and α-fructose
α-fructose and α-galactose
α-glucose and α-galactose
Name the three digestible disaccharides we talked about. What monosaccharides are they each made of and tell whether they are connected by an alpha 1-4 glycosidic linkage, a 1-4 beta glycosidic linkage or a 1-5 beta glycosidic linkage.
Chapter 5 Solutions
Study Guide for Campbell Biology
Ch. 5 - Monomers are linked into polymers by ________...Ch. 5 - You can recognize a monosaccharide by its multiple...Ch. 5 - Number the carbons in the following glucose and...Ch. 5 - Prob. 4IQCh. 5 - Fill in this concept map to help you organize your...Ch. 5 - a. Draw the amino acids alanine (R group: CH3) and...Ch. 5 - In the following diagram of a portion of a...Ch. 5 - Now that you have gained experience with concept...Ch. 5 - a. Label the three parts of this nucleotide....Ch. 5 - Take the time to create a concept map that...
Ch. 5 - Prob. 1SYKCh. 5 - Prob. 2SYKCh. 5 - glycogen A. carbohydrate B. lipid C. protein D....Ch. 5 - cholesterol A. carbohydrate B. lipid C. protein D....Ch. 5 - RNA A. carbohydrate B. lipid C. protein D. nucleic...Ch. 5 - collagen A. carbohydrate B. lipid C. protein D....Ch. 5 - hemoglobin A. carbohydrate B. lipid C. protein D....Ch. 5 - A. carbohydrate B. lipid C. protein D. nucleic...Ch. 5 - Prob. 7TYKMCh. 5 - enzyme A. carbohydrate B. lipid C. protein D....Ch. 5 - cellulose A. carbohydrate B. lipid C. protein D....Ch. 5 - Chitin A. carbohydrate B. lipid C. protein D....Ch. 5 - Polymerization (the formation of polymers) is a...Ch. 5 - Which of the following statements is not true of a...Ch. 5 - Prob. 3TYKCh. 5 - Prob. 4TYKCh. 5 - Prob. 5TYKCh. 5 - Prob. 6TYKCh. 5 - A fatty acid that has the formula C16H32O2 is a....Ch. 5 - Prob. 8TYKCh. 5 - Prob. 9TYKCh. 5 - Prob. 10TYKCh. 5 - Which of the following molecules provides the most...Ch. 5 - Prob. 12TYKCh. 5 - What happens when a protein denatures? a. Its...Ch. 5 - The helix of proteins is a. part of a proteins...Ch. 5 - What is the best description of the following...Ch. 5 - Prob. 16TYKCh. 5 - Prob. 17TYKCh. 5 - Which of the following is true of the subunits of...Ch. 5 - Prob. 19TYKCh. 5 - If the nucleotide sequence of one strand of a DNA...Ch. 5 - How are nucleotide monomers connected to form a...Ch. 5 - Prob. 22TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- draw the following amino acid chains and give the single and three letter abbreviations for cysteinearrow_forwardDescribe the common structural features and the differences for eachof the following pairs: (a) cellulose and glycogen; (b) D-glucose and D-fructose; (c) maltose and sucrose.arrow_forwardDraw the structure and give the systematic name of the given disaccharides. Lactose which is also known as milk sugar: β-D-Gal(1à4)β-D-Glc, where Gal is galactose and Glc is glucose. Sucrose which is also known as table sugar: a-D-Glc (1-->2) b-D-Fru where Glc is glucose and Fru is fructose.arrow_forward
- An amylose chain is 5000 glucose units long. At how many places must it be cleaved to reduce the average length to 2500 units? To 1000 units? To 200 units? What percentage of the glycosidic links are hydrolyzed in each case? (Even partial hydrolysis can drastically alter the physical properties of polysaccharides and thus affect their structural role in )arrow_forwardSugars are technically called carbohydrates, referring to the fact that their formulae are only multiple of C(H₂O). Pentoses therefore have five carbons, ten hydrogens and five oxygen atoms. Ribose is a pentose. Choose from among the following another pentose. A. Fructose B. Ribulose C. Galactose D. Erythrose Monomers are bonded together by which of the following processes? A. Hydrolysis B. Non-hydration lysis C. Ionic Bonding D. Dehydration Synthesisarrow_forwardAssume you could make a trisaccharide by covalently joining xylose (C5H10O5) , xylose (C5H10O5) and fructose (C6H12O6). So 2x xylose, 1x fructose. What would be the chemical formula of this trisaccharide assuming regular dehydration synthesis as discussed in class? You do not need to worry about which carbons are being used for forming these glycosidic linkages.arrow_forward
- A student decided that the configuration of the asymmetric centers in a sugar such as d-glucose could be determined rapidly by simply assigning the Rconfiguration to an asymmetric center with an OH group on the right and the S configuration to an asymmetric center with an OH group on the left. Ishe correct? (We will see in Chapter 20 that the “d” in d-glucose means that the OH group on the bottommost asymmetric center is on the right.)arrow_forwardWhy is lactose a reducing sugar while sucrose is a non-reducing sugar? Explain clearly. Please support the answer with an illustration.arrow_forwardName which, if any, of the following are epimers ofd-glucose: d-mannose, d-galactose, d-ribosearrow_forward
- Name which, if any, of the following are epimers of d-glucose: Dmannose, D-galactose, D-ribose.arrow_forward1) Please list all glycosidic linkages between each monosaccharide units. For example, α(1→4)2) Please discuss whether these oligo/polysaccharides would be reducing or non-reducing sugar. Remember to state your reasoning in complete sentence.arrow_forwardDraw the respective enantiomer of the following sugar molecules: D-glyceraldehyde D-glucose D-mannose D-fructosearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license