
One reaction that occurs in human
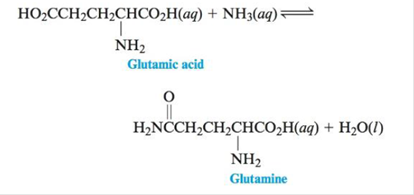
For this reaction ∆G°= 14 kJ at 25°c.
a. Calculate K for this reaction at 25°C.
b. In a living cell this reaction is coupled with the hydrolysis of ATP. (See Exercise 75.) Calculate ∆G° and K at 25°C for the following reaction:
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Chemistry: An Atoms First Approach
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: The Central Science (13th Edition)
Organic Chemistry - Standalone book
Chemistry & Chemical Reactivity
Chemistry by OpenStax (2015-05-04)
- Which contains greater entropy, a quantity of frozen benzene or the same quantity of liquid benzene at the same temperature? Explain in terms of the dispersal of energy in the substance.arrow_forwardTetrachloromethane (carbon tetrachloride), CCl4, has a normal boiling point of 76.7C and an enthalpy of vaporization, Hvap, of 29.82 kJ/mol. Estimate the entropy of vaporization, Svap. Estimate the free energy of vaporization, Gvap, at 25C.arrow_forwardFor the reaction CH3OH(l)+32O2(g)2H2O(l)+CO2(g) the value of G is 702.2 kJ at 25C. Other data are as follows: Hf (kJ/mol) at 25C S (J/molK) at 25C CH3OH(l) 238.7 126.8 H2O(l) 285.8 70.0 CO2(g) 393.5 213.7 Calculate the standard entropy, S, per mole of O2(g).arrow_forward
- For the decomposition of formic acid, HCOOH(l)H2O(l)+CO(g) H = +29 kJ/mol at 25C. a Does the tendency of this reaction to proceed to a state of minimum energy favor the formation of water and carbon monoxide or formic acid? Explain. b Does the tendency of this reaction to proceed to a state of maximum entropy favor the formation of products or reactants? Explainarrow_forwardDiethyl ether, (C2H5)2O, was once used as an anesthetic. Calculate the entropy change, rS, for the vaporization of ether if its heat of vaporization is 26.0 kJ/mol at the boiling point of 35.0 C.arrow_forwardHuman DNA contains almost twice as much information as is needed to code for all the substances produced in the body. Likewise, the digital data sent from Voyager II contained one redundant bit out of every two bits of information. The Hubble space telescope transmits three redundant bits for every bit of information. How is entropy related to the transmission of information? What do you think is accomplished by having so many redundant bits of information in both DNA and the space probes?arrow_forward
- According to Lambert, leaves lying in the yard and playing cards that are in disarray on a table have not undergone an increase in their thermodynamic entropy. Suggest another reason why leaves and playing cards may not be a good analogy for the entropy of a system containing, for example, only H2O molecules or only O2 molecules.arrow_forwardConsider the reaction of 2 mol H2(g) at 25C and 1 atm with 1 mol O2(g) at the same temperature and pressure to produce liquid water at these conditions. If this reaction is run in a controlled way to generate work, what is the maximum useful work that can be obtained? How much entropy is produced in this case?arrow_forwardConsider the reaction of 1 mol H2(g) at 25C and 1 atm with 1 mol Br2(l) at the same temperature and pressure to produce gaseous HBr at these conditions. If this reaction is run in a controlled way to generate work, what is the maximum useful work that can be obtained? How much entropy is produced in this case?arrow_forward
- Explain why each of the following statements is incorrect. (a) Entropy increases in all spontaneous reactions. (b) Reactions with a negative free energy change (rG 0) are product-favored and occur with rapid transformation of reactants to products. (c) All spontaneous processes are exothermic. (d) Endothermic processes are never spontaneous.arrow_forwardIn muscle cells under the condition of vigorous exercise, glucose is converted to lactic acid (lactate),CH3CHOHCOOH, by the chemical reaction C6H12O6 2 CH3CHOHCOOHrG = 197 kJ/mol (a) If all of the Gibbs free energy from this reaction wereused to convert ADP to ATP, calculate how many molesof ATP could be produced per mole of glucose. (b) The actual reaction involves the production of 3 molATP per mole of glucose. Calculate the rG for thisoverall reaction. (c) Is the overall reaction in part (b) reactant-favored orproduct-favored?arrow_forwardWhat is entropy? Why is entropy important?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





