
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 94AE
Consider the following diagram of free energy (G) versus fraction of A reacted in terms of moles for the reaction 2A(g) →B(g).
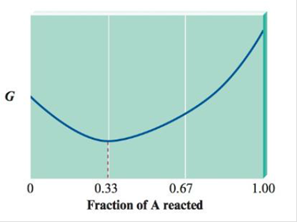
Before any A has reacted, PA = 3.0 atm and PB = 0. Determine the sign of ∆G° and the value of Kp. for this reaction.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 16 Solutions
Chemistry: An Atoms First Approach
Ch. 16 - Define the following: a. spontaneous process b....Ch. 16 - What is the second law of thermodynamics? For any...Ch. 16 - Prob. 3RQCh. 16 - Prob. 4RQCh. 16 - Prob. 5RQCh. 16 - What is the standard free energy change, G, for a...Ch. 16 - If you calculate a value for G for a reaction...Ch. 16 - Consider the equation G = G + RT ln(Q). What is...Ch. 16 - Even if G is negative, the reaction may not occur....Ch. 16 - Prob. 10RQ
Ch. 16 - For the process A(l) A(g), which direction is...Ch. 16 - Prob. 2ALQCh. 16 - Gas A2 reacts with gas B2 to form gas AB at a...Ch. 16 - Prob. 4ALQCh. 16 - Prob. 5ALQCh. 16 - Prob. 6ALQCh. 16 - Predict the sign of S for each of the following...Ch. 16 - Is Ssurr favorable or unfavorable for exothermic...Ch. 16 - At 1 atm, liquid water is heated above 100C. For...Ch. 16 - Prob. 10ALQCh. 16 - The synthesis of glucose directly from CO2 and H2O...Ch. 16 - When the environment is contaminated by a toxic or...Ch. 16 - Entropy has been described as times arrow....Ch. 16 - Prob. 14QCh. 16 - A mixture of hydrogen gas and chlorine gas remains...Ch. 16 - Consider the following potential energy plots: a....Ch. 16 - Prob. 17QCh. 16 - Given the following illustration, what can be said...Ch. 16 - The third law of thermodynamics states that the...Ch. 16 - Prob. 20QCh. 16 - Prob. 21QCh. 16 - Prob. 22QCh. 16 - Monochloroethane (C2H5Cl) can be produced by the...Ch. 16 - Prob. 24QCh. 16 - Which of the following processes are spontaneous?...Ch. 16 - Which of the following processes are spontaneous?...Ch. 16 - Prob. 27ECh. 16 - Consider the following illustration of six...Ch. 16 - Consider the following energy levels, each capable...Ch. 16 - Prob. 30ECh. 16 - Choose the substance with the larger positional...Ch. 16 - Which of the following involve an increase in the...Ch. 16 - Predict the sign of Ssurr for the following...Ch. 16 - Prob. 34ECh. 16 - Given the values of H and S, which of the...Ch. 16 - At what temperatures will the following processes...Ch. 16 - Ethanethiol (C2H5SH; also called ethyl mercaptan)...Ch. 16 - For mercury, the enthalpy of vaporization is 58.51...Ch. 16 - For ammonia (NH3), the enthalpy of fusion is 5.65...Ch. 16 - The enthalpy of vaporization of ethanol is 38.7...Ch. 16 - Predict the sign of S for each of the following...Ch. 16 - Prob. 42ECh. 16 - Prob. 43ECh. 16 - For each of the following pairs, which substance...Ch. 16 - Predict the sign of S and then calculate S for...Ch. 16 - Predict the sign of S and then calculate S for...Ch. 16 - Prob. 47ECh. 16 - Prob. 48ECh. 16 - Prob. 49ECh. 16 - Two crystalline forms of white phosphorus are...Ch. 16 - Consider the reaction 2O(g)O2(g) a. Predict the...Ch. 16 - Prob. 52ECh. 16 - Prob. 53ECh. 16 - The major industrial use of hydrogen is in the...Ch. 16 - Prob. 55ECh. 16 - At 100C and 1.00 atm, H = 40.6 kJ/mol for the...Ch. 16 - Prob. 57ECh. 16 - Prob. 58ECh. 16 - Prob. 59ECh. 16 - Prob. 60ECh. 16 - Consider the reaction...Ch. 16 - Consider the reaction 2POCl3(g)2PCl3(g)+O2(g) a....Ch. 16 - Prob. 63ECh. 16 - Consider two reactions for the production of...Ch. 16 - Prob. 65ECh. 16 - Prob. 66ECh. 16 - Consider the reaction 2NO2(g)N2O4(g) For each of...Ch. 16 - Prob. 68ECh. 16 - One of the reactions that destroys ozone in the...Ch. 16 - Hydrogen sulfide can be removed from natural gas...Ch. 16 - Consider the following reaction at 25.0C:...Ch. 16 - The standard free energies of formation and the...Ch. 16 - Calculate G forH2O(g)+12O2(g)H2O2(g) at 600. K,...Ch. 16 - The Ostwald process for the commercial production...Ch. 16 - Cells use the hydrolysis of adenosine...Ch. 16 - One reaction that occurs in human metabolism is...Ch. 16 - Prob. 77ECh. 16 - Consider the following reaction at 298 K:...Ch. 16 - Prob. 79ECh. 16 - The equilibrium constant K for the reaction...Ch. 16 - Prob. 81AECh. 16 - Some water is placed in a coffee-cup calorimeter....Ch. 16 - Consider the following system at equilibrium at...Ch. 16 - Calculate the entropy change for the vaporization...Ch. 16 - As O2(l) is cooled at 1 atm, it freezes at 54.5 K...Ch. 16 - Prob. 86AECh. 16 - Using the following data, calculate the value of...Ch. 16 - Prob. 88AECh. 16 - Carbon monoxide is toxic because it bonds much...Ch. 16 - Prob. 90AECh. 16 - Prob. 91AECh. 16 - Use the equation in Exercise 79 to determine H and...Ch. 16 - Consider the reaction...Ch. 16 - Consider the following diagram of free energy (G)...Ch. 16 - Prob. 95CWPCh. 16 - For rubidium Hvapo=69.0KJ/mol at 686C, its boiling...Ch. 16 - Given the thermodynamic data below, calculate S...Ch. 16 - Prob. 98CWPCh. 16 - Prob. 99CWPCh. 16 - Consider the dissociation of a weak acid HA (Ka =...Ch. 16 - Prob. 101CWPCh. 16 - The equilibrium constant for a certain reaction...Ch. 16 - For the following reactions at constant pressure,...Ch. 16 - The standard enthalpy of formation of H2O(l) at...Ch. 16 - Prob. 105CPCh. 16 - Liquid water at 25C is introduced into an...Ch. 16 - Using data from Appendix 4, calculate H, G, and K...Ch. 16 - Prob. 108CPCh. 16 - Prob. 109CPCh. 16 - Prob. 110CPCh. 16 - Prob. 111CPCh. 16 - Prob. 112CPCh. 16 - If wet silver carbonate is dried in a stream of...Ch. 16 - Carbon tetrachloride (CCl4) and benzene (C6H6)...Ch. 16 - Sodium chloride is added to water (at 25C) until...Ch. 16 - Prob. 116CPCh. 16 - Prob. 117CPCh. 16 - Prob. 118IPCh. 16 - Prob. 119IPCh. 16 - Prob. 120IPCh. 16 - Consider a sample containing 5.00 moles of a...Ch. 16 - Impure nickel, refined by smelting sulfide ores in...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The free energy for a reaction decreases as temperature increases. Explain how this observation is used to determine the sign of either H or S.arrow_forwardWhat is the sign of the standard Gibbs free-energy change at low temperatures and at high temperatures for the synthesis of ammonia? 3H2(g) + N2(g) 2NH3(g)arrow_forwardAt 298 K, G = 70.52 kJ for the reaction 2NO(g) + O2(g) 2NO2(g) (a) Calculate _G at the same temperature when PNO = 1.0 104 atm, PO2=2.0103 atm, and PNO2=0.30 atm. (b) Under the conditions in part a, in which direction is the reaction spontaneous?arrow_forward
- Consider the reaction of 1 mol H2(g) at 25C and 1 atm with 1 mol Br2(l) at the same temperature and pressure to produce gaseous HBr at these conditions. If this reaction is run in a controlled way to generate work, what is the maximum useful work that can be obtained? How much entropy is produced in this case?arrow_forwardConsider the reaction of 2 mol H2(g) at 25C and 1 atm with 1 mol O2(g) at the same temperature and pressure to produce liquid water at these conditions. If this reaction is run in a controlled way to generate work, what is the maximum useful work that can be obtained? How much entropy is produced in this case?arrow_forwardChemists and engineers who design nuclear power plants have to worry about high-temperature reactions because it is possible for water to decompose. (a) Under what conditions does this reaction occur spontaneously? 2H2O(g) 2H2(g) + O2(g) (b) Under conditions where the decomposition of water is spontaneous, do nuclear engineers have to worry about an oxygen/hydrogen explosion? Justify your answer.arrow_forward
- Yeast can produce ethanol by the fermentation of glucose (C6H12O6), which is the basis for the production of most alcoholic beverages. C6H12O6(aq) 2 C2H5OH() + 2 CO2(g) Calculate rH, rS, and rG for the reaction at 25 C. Is the reaction product- or reactant-favored at equilibrium? In addition to the thermodynamic values in Appendix L, you will need the following data for C6H12O6(aq): fH = 1260.0 kl/mol; S = 289 J/K mol; and fG = 918.8 kl/mol.arrow_forwardFor each process, predict whether entropy increases or decreases, and explain how you arrived at your prediction. 2 CO2(g) → 2 CO(g) + O2(g) NaCl(s) → NaCl(aq) MgCO3(s) → MgO(s) + CO2(g)arrow_forwardWhat is the third law of thermodynamics? What are standard entropy values, S, and how are these S values (listed in Appendix 4) used to calculate S for a reaction? How would you use Hesss law to calculate S for a reaction? What does the superscript indicate? Predicting the sign of S for a reaction is an important skill to master. For a gas-phase reaction, what do you concentrate on to predict the sign of S? For a phase change, what do you concentrate on to predict the sign of S? That is, how are Ssolid, Sliquid, and Sgas related to one another? When a solute dissolves in water, what is usually the sign of S for this process?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY