
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 21PS
Consider the following half-reactions:
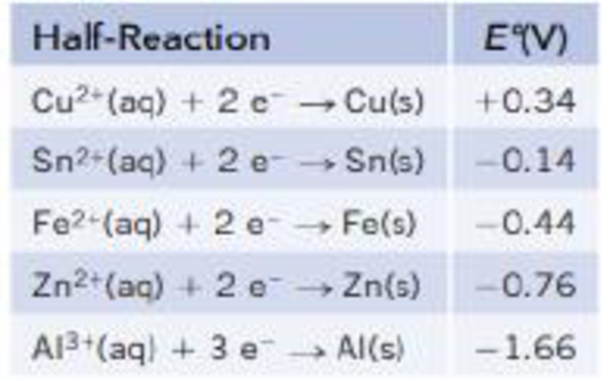
- (a) Based on E° values, which metal is the most easily oxidized?
- (b) Which metals on this list are capable of reducing Fe2+(aq) to Fe(s)?
- (c) Write a balanced chemical equation tor the reaction of Fe2+(aq) with Sn(s). Is this reaction product-favored or reactant-favored at equilibrium?
- (d) Write a balanced chemical equation for the reaction of Zn2+ (aq) with Sn(s). Is this reaction product-favored or react ant-favored at equilibrium?
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 19 Solutions
Chemistry & Chemical Reactivity
Ch. 19.1 - A common laboratory analysis for iron is to...Ch. 19.1 - Prob. 19.2CYUCh. 19.2 - Describe how to set up a voltaic cell using the...Ch. 19.2 - The following overall chemical reaction occurs in...Ch. 19.4 - (a) Rank the following metals in their ability to...Ch. 19.5 - A voltaic cell is set up with an aluminum...Ch. 19.5 - The half-cells Ag+(aq. 1.0 M)|Ag(s) and H+(aq, ?...Ch. 19.6 - Prob. 19.8CYUCh. 19.6 - Calculate the equilibrium constant at 25 C for the...Ch. 19.7 - Predict the chemical reactions that will occur at...
Ch. 19.8 - Prob. 19.11CYUCh. 19.9 - Prob. 1.1ACPCh. 19.9 - Prob. 1.2ACPCh. 19.9 - Prob. 1.3ACPCh. 19.9 - Prob. 2.1ACPCh. 19.9 - Use standard reduction potentials to determine...Ch. 19.9 - Prob. 2.3ACPCh. 19.9 - The overall reaction for the production of Cu(OH)2...Ch. 19.9 - Assume the following electrochemical cell...Ch. 19 - Write balanced equations for the following...Ch. 19 - Write balanced equations for the following...Ch. 19 - Balance the following redox equations. All occur...Ch. 19 - Balance the following redox equations. All occur...Ch. 19 - Balance the following redox equations. All occur...Ch. 19 - Prob. 6PSCh. 19 - A voltaic cell is constructed using the reaction...Ch. 19 - A voltaic cell is constructed using the reaction...Ch. 19 - The half-cells Fe2+(aq) | Fe(s) and O2(g) | H2O...Ch. 19 - The half cells Sn2+(aq) |Sn(s) and Cl2(g) |Cl(aq)...Ch. 19 - For each of the following electrochemical cells,...Ch. 19 - For each of the following electrochemical cells,...Ch. 19 - Use cell notation to depict an electrochemical...Ch. 19 - Use cell notation to depict an electrochemical...Ch. 19 - What are the similarities and differences between...Ch. 19 - What reactions occur when a lead storage battery...Ch. 19 - Calculate the value of E for each of the following...Ch. 19 - Calculate the value of E for each of the following...Ch. 19 - Balance each of the following unbalanced...Ch. 19 - Balance each of the following unbalanced...Ch. 19 - Consider the following half-reactions: (a) Based...Ch. 19 - Prob. 22PSCh. 19 - Which of the following elements is the best...Ch. 19 - Prob. 24PSCh. 19 - Which of the following ions is most easily...Ch. 19 - From the following list, identify the ions that...Ch. 19 - (a) Which halogen is most easily reduced in acidic...Ch. 19 - Prob. 28PSCh. 19 - Calculate the potential delivered by a voltaic...Ch. 19 - Calculate the potential developed by a voltaic...Ch. 19 - One half-cell in a voltaic cell is constructed...Ch. 19 - One half-cell in a voltaic cell is constructed...Ch. 19 - One half-cell in a voltaic cell is constructed...Ch. 19 - One half-cell in a voltaic cell is constructed...Ch. 19 - Calculate rG and the equilibrium constant for the...Ch. 19 - Prob. 36PSCh. 19 - Use standard reduction potentials (Appendix M) for...Ch. 19 - Use the standard reduction potentials (Appendix M)...Ch. 19 - Use the standard reduction potentials (Appendix M)...Ch. 19 - Use the standard reduction potentials (Appendix M)...Ch. 19 - Prob. 41PSCh. 19 - Prob. 42PSCh. 19 - Which product, O2 or F2, is more likely to form at...Ch. 19 - Which product, Ca or H2, is more likely to form at...Ch. 19 - An aqueous solution of KBr is placed in a beaker...Ch. 19 - An aqueous solution of Na2S is placed in a beaker...Ch. 19 - In the electrolysis of a solution containing...Ch. 19 - In the electrolysis of a solution containing...Ch. 19 - Electrolysis of a solution of CuSO4(aq) to give...Ch. 19 - Electrolysis of a solution of Zn(NO3)2(aq) to give...Ch. 19 - A voltaic cell can be built using the reaction...Ch. 19 - Assume the specifications of a Ni-Cd voltaic cell...Ch. 19 - Use E values to predict which of the following...Ch. 19 - Prob. 54PSCh. 19 - Prob. 55PSCh. 19 - Prob. 56PSCh. 19 - Prob. 57GQCh. 19 - Balance the following equations. (a) Zn(s) +...Ch. 19 - Magnesium metal is oxidized, and silver ions are...Ch. 19 - You want to set up a series of voltaic cells with...Ch. 19 - Prob. 61GQCh. 19 - Prob. 62GQCh. 19 - In the table of standard reduction potentials,...Ch. 19 - Prob. 64GQCh. 19 - Four voltaic cells are set up. In each, one...Ch. 19 - The following half-cells are available: (i)...Ch. 19 - Prob. 67GQCh. 19 - Prob. 68GQCh. 19 - A potential of 0.142 V is recorded (under standard...Ch. 19 - Prob. 70GQCh. 19 - The standard potential, E, for the reaction of...Ch. 19 - An electrolysis cell for aluminum production...Ch. 19 - Electrolysis of molten NaCl is done in cells...Ch. 19 - A current of 0.0100 A is passed through a solution...Ch. 19 - A current of 0.44 A is passed through a solution...Ch. 19 - Prob. 76GQCh. 19 - Prob. 77GQCh. 19 - Prob. 78GQCh. 19 - The products formed in the electrolysis of aqueous...Ch. 19 - Predict the products formed in the electrolysis of...Ch. 19 - Prob. 81GQCh. 19 - The metallurgy of aluminum involves electrolysis...Ch. 19 - Prob. 83GQCh. 19 - Prob. 84GQCh. 19 - Prob. 85GQCh. 19 - Prob. 86GQCh. 19 - Two Ag+(aq) | Ag(s) half-cells are constructed....Ch. 19 - Calculate equilibrium constants for the following...Ch. 19 - Prob. 89GQCh. 19 - Use the table of standard reduction potentials...Ch. 19 - Prob. 91GQCh. 19 - Prob. 92GQCh. 19 - Prob. 93GQCh. 19 - A voltaic cell is constructed in which one...Ch. 19 - An expensive but lighter alternative to the lead...Ch. 19 - The specifications for a lead storage battery...Ch. 19 - Manganese may play an important role in chemical...Ch. 19 - Prob. 98GQCh. 19 - Iron(II) ion undergoes a disproportionation...Ch. 19 - Copper(I) ion disproportionates to copper metal...Ch. 19 - Prob. 101GQCh. 19 - Prob. 102GQCh. 19 - Can either sodium or potassium metal be used as a...Ch. 19 - Galvanized steel pipes are used in the plumbing of...Ch. 19 - Consider an electrochemical cell based on the...Ch. 19 - Prob. 106ILCh. 19 - A silver coulometer (Study Question 106) was used...Ch. 19 - Four metals, A, B, C, and D, exhibit the following...Ch. 19 - Prob. 109ILCh. 19 - The amount of oxygen, O2, dissolved in a water...Ch. 19 - Prob. 111SCQCh. 19 - The free energy change for a reaction, rG, is the...Ch. 19 - Prob. 113SCQCh. 19 - (a) Is it easier to reduce water in acid or base?...Ch. 19 - Prob. 115SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- An aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.39 hours. The electroplating is carried out with an efficiency of 93.0%, resulting in a deposit of 21.221 g of gold. a How many faradays are required to deposit the gold? b What is the charge on the gold ions (based on your calculations)?arrow_forwardA lead storage battery delivers a current of 6.00 A for one hour and 22 minutes at a voltage of 12.0 V. (a) How many grams of lead are converted to PbSO4? (b) How much electrical energy is produced in kilowatt hours?arrow_forwardA solution contains the ions H+, Ag+, Pb2+, and Ba2+, each at a concentration of 1.0 M. (a) Which of these ions would be reduced first at the cathode during an electrolysis? (b) After the first ion has been completely removed by electrolysis, which is the second ion to be reduced? (c) Which, if any, of these ions cannot be reduced by the electrolysis of the aqueous solution?arrow_forward
- The black silver sulfide discoloration of silverware can be removed by heating the silver article in a sodium carbonate solution in an aluminum pan. The reaction is 3Ag2S(s)+2Al(s)6Ag(s)+3S2(aq)+2Al3+(aq) a. Using data in Appendix 4, calculate G, K, and for the above reaction at 25C. [For Al3+(aq), Gf = 480. kJ/mol.] b. Calculate the value of the standard reduction potential for the following half-reaction: 2e+Ag2S(s)2Ag(s)+S2(aq)arrow_forwardChlorine, Cl2, is produced commercially by the electrolysis of aqueous sodium chloride. The anode reaction is 2Cl(aq)Cl2(g)+2e How long will it take to produce 2.00 kg of chlorine if the current is 5.00 102 A?arrow_forwardAn electrolysis experiment is performed to determine the value of the Faraday constant (number of coulombs per mole of electrons). In this experiment, 28.8 g of gold is plated out from a AuCN solution by running an electrolytic cell for two hours with a current of 2.00 A. What is the experimental value obtained for the Faraday Constant?arrow_forward
- When copper reacts with nitric acid, a mixture of NO(g) and NO2(g) is evolved. The volume ratio of the two product gases depends on the concentration of the nitric acid according to the equilibrium 2H+(aq)+2NO3(aq)+NO(g)3NO2(g)+H2O(l) Consider the following standard reduction potentials at 25C: a. Calculate the equilibrium constant for the above reaction. b. What concentration of nitric acid will produce a NO and NO2 mixture with only 0.20% NO2 (by moles) at 25C and 1.00 atm? Assume that no other gases are present and that the change in acid concentration can be neglected.arrow_forwardCalculate the maximum work available from 50.0 g of aluminum in the following cell when the cell potential is 1.15 V. Al(s)Al3+(aq)H+(aq)O2(g)Pt Note that O2 is reduced to H2O.arrow_forwardCalculate the standard cell potential of the cell corresponding to the oxidation of oxalic acid, H2C2O4, by permanganate ion. MnO4. 5H2C2O4(aq)+2MnO4(aq)+6H+(aq)10CO2(g)+2Mn2+(aq)+8H2O(l) See Appendix C for free energies of formation: Gf for H2C2O4(aq) is 698 kJ.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Electrolysis; Author: Tyler DeWitt;https://www.youtube.com/watch?v=dRtSjJCKkIo;License: Standard YouTube License, CC-BY