
(a)
Interpretation:
The curved-arrow mechanism for the anionic oxyCope reaction of compound A is to be given and the stereoisomer B does not react under the same conditions-the statement is to be explained.
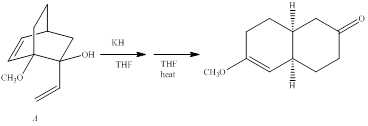
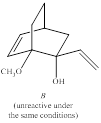
Concept introduction:
Generally, [3, 3] sigmatropic reactions of 1, 5-diences are called as cope reactions. When the diene is having the oxygen at C3 or C4 position the sigmatropic reaction is called as oxy cope reaction. Notably, when the alkoxide fragment is involved it is referred as anionic oxy cope reaction. Moreover, the enhanced increase in
(b)
Interpretation:
The structure of the product, including its stereochemistry expected from the anion oxyCope reaction of the following compound is to be given.

Concept introduction:
When the diene is having the oxygen at C3 or C4 position the sigmatropic reaction is called as oxy cope reaction. Notably, when the alkoxide fragment is involved it is referred as anionic oxy cope reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid gave B which on reduction with LiAH4 gave C. Oxidation of compound C with chromic acid gave D (C9H14O). Suggest the structures for A, B, C, and D.arrow_forwardCompound A is an alcohol that undergoes oxidation to produce compound B.Compound B is a ketone that gives positive triiodomethane reaction. Compound B isthen reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueousacid to form compound C. Compound C has the molecular formula of C9H12O. Deducethe structure for compound A, B and C. PLEASE PROVIDE CLEAR DRAWINGS AND EXPLANATIONSarrow_forwardGive the structural chemistry of active methylene, classify it asradical/intermediate/stable organic compound, reagent of its production and role in alkylationarrow_forward
- Give the structure of the product and/or intermediates of the following reactions. Indicate, whenappropriate, both regiochemistry and stereochemistryarrow_forwardthe following reaction scheme leads to the formation of compound B. give the structure of the final products and of the intermediate product A and justify, using the mechanism, the formation of thesearrow_forwardGive the major organic product of the following reaction for a) or b)arrow_forward
- (b) Classify the following alcohols in order of increasing ease of acid-catalyzed dehydration.Justify your answers.arrow_forward3. Obtain acetophenone and acetaldehyde by reaction of glycols with periodic acid. Justify your answer with the reaction mechanism.arrow_forwardGive the process of Determining the Mechanism and Stereochemistry inNucleophilic Substitution ?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
