
(a)
Interpretation:
Each of the following reactions involves a sequence of two pericyclic reactions. The intermediate X involved in each reaction, and pericyclic reactions involved are to be depicted.
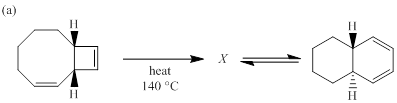
Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner. In phase orbital overlap results in symmetry allowed electrocyclic reactions. Selection rules of electrocyclic reactions are;
| No. of electrons | Activation mode | Stereochemistry of rotation |
| 4n | Thermal Photochemical |
Con Dis |
| 4n + 2 | Thermal Photochemical |
Dis Con |
(b)
Interpretation:
Each of the following reactions involves a sequence of two pericyclic reactions. The intermediate Y involved in each reaction, and pericyclic reactions involved are to be depicted.
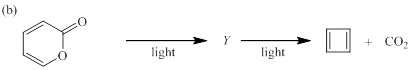
Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- The transformation takes place via two sequential pericyclic reactions. Identify the two reactions and give a critical explanation whether the reactions are allowable or not. Explain the stereochemistry.arrow_forwardPredict the product(s) of the following reactions, including stereochemistry when necessary and identify the mechanism of each substitution reaction (SN1 vs SN2). Draw the reaction mechanism (reaction arrows) for any one of the reactions to show how the product is formed.arrow_forwardwhat are the products? please indicate the relative stereochemistry for the following transformations and indicate the major product.arrow_forward
- What is the major substitution product for the following reaction? Show the mechanism for the reaction.arrow_forwardFor each one of the following reactions, show step by step mechanism of the reactions and indicate the stereochemistry of the products and their relationshiparrow_forwardIdentify the major product and the mechanism involved:arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

