
Concept explainers
(a)
Interpretation:
Each of the following compounds exists as a fluxional molecule that is interconverted into one or more identical forms by the sigmatropic process indicated. One structure in each case that demonstrates the process involved is to be drawn. Each process is an allowed pericyclic reaction – the statement is to be verified.
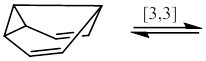
Concept introduction:
When a molecule undergoes a dynamic molecular motion involving the interchange of two or more groups which are chemically and magnetically different, then the molecule is said to have fluxionality nature. In this case, the signals corresponding to interchanged two or more groups will be identical, if the rate of exchange of the groups occurs very quickly. The fluxionality in organic molecules can be easily followed by multinuclear NMR spectroscopy.
(b)
Interpretation:
Each of the following compounds exists as a fluxional molecule that is interconverted into one or more identical forms by the sigmatropic process indicated. One structure in each case that demonstrates the process involved is to be drawn. Each process is an allowed pericyclic reaction – the statement is to be verified.
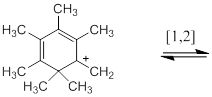
Concept introduction:
When a molecule undergoes a dynamic molecular motion involving the interchange of two or more groups which are chemically and magnetically different, then the molecule is said to have fluxionality nature. In this case, the signals corresponding to interchanged two or more groups will be identical, if the rate of exchange of the groups occurs very quickly. The fluxionality in organic molecules can be easily followed by multinuclear NMR spectroscopy.
Trending nowThis is a popular solution!

Chapter 28 Solutions
Organic Chemistry
- Since compounds A and B are in different comformations, how do the products of the elimation reaction differ in conformation between these two compounds? Please provide visual drawing to explain.arrow_forwardWrite down the explicit structures of A, B, C and D in accordance with the reactions given in the synthesis plan, which shows the synthesis of D, a pheromone of moth.arrow_forwardIdentify the pericyclic reactions in the followingreaction schemes. Give the complete reactionname and indicate the course of the reaction withthe aid of the arrow notation.arrow_forward
- For the following reactions:a. Provide the missing major product, OR, missing reagent and conditions appropriate.b. Pay attention to stereochemistry unless directed otherwise and identify any racemicmixture using the symbol (±)c. Assign each reaction as addition, elimination, substitution or rearrangement, unlessdirected otherwise.arrow_forwardPredict the major organic product of the following reaction for a and or barrow_forwardGive a clear handwritten answer with explanation....give also textual explanation..give the name of product with explanation and give also product stereochemistry...?arrow_forward
- Explain why compound A will not undergo a ring-opening reaction under thermal conditions, but compound B will.arrow_forwardGive the major organic product of the following reaction for a) or b)arrow_forwardwith the help of symmetry properties of molecular orbitals of cyclohexadiene ,show why its con-rotatory conversion to hexatriene is thermally forbidden processarrow_forward
- Suggest reactivity of compound A, B and C in increasing order of E2 reactionarrow_forwardGive the MAJOR product(s) for the following reactions. For example, if BOTHelimination and substitution would occur, write both products. Or, if two carboncontaining groups would be formed as a result of a molecule splitting, write bothorganic molecules. Also, as indicated, describe the stereochemistry of theproducts. If no stereochemistry exists in the final product, but if astereochemistry answer box is included for that problem, then write NOSTEREOCHEMISTRY, NONE or N/A in the corresponding box. At the end, youwill be asked to label which problems yielded products that were examples ofhemiacetals/ acetals, hemiketals/ketals, and Schiff bases. There is onehemiacetal/acetal or hemi-ketal/ketal, and one Schiff base.arrow_forwardGive the MAJOR product(s) for the following reactions. For example, if BOTHelimination and substitution would occur, write both products. Or, if two carboncontaining groups would be formed as a result of a molecule splitting, write bothorganic molecules. Also, as indicated, describe the stereochemistry of theproducts. If no stereochemistry exists in the final product, but if astereochemistry answer box is included for that problem, then write NOSTEREOCHEMISTRY, NONE or N/A in the corresponding box. At the end, youwill be asked to label which problems yielded products that were examples ofhemiacetals/ acetals, hemiketals/ketals, and Schiff bases. There is onehemiacetal/acetal or hemi-ketal/ketal, and one Schiff base. Do them in structural format given and show you work step by steparrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

