
(a)
Interpretation:
The transformation shown in Fig. P28.50, which involves a sequence of two pericyclic reactions, was used as a key step in a synthesis of the sex hormone estrone. The unstable intermediate A is to be identified and the mechanism for both its formation and its subsequent reaction is to be stated.

Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner.
(b)
Interpretation:
Conversion of product of part (a) into estrone is to be stated.
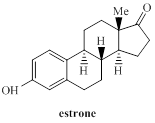
Concept introduction:
The alcohols (primary and secondary) can be oxidized by using the oxidizing agents like Pyridinium chlorochromate (PCC) and Pyridinium dichromate (PDC).Similarly, demethylation reaction can be accomplished by using HI in heating conditions or by using boron tribromide in dichloromethane.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- The ketone shown was prepared in a three-step sequence from ethyl trifluoroacetate. The first step in the sequence involved treating ethyl trifluoroacetate with ammonia to give compound A. Compound A was in turn converted to the desired ketone by way of compound B. Fill in the missing reagents in the sequence shown, and give the structures of compounds A and B.arrow_forwardDeduce the structures of compound A,B,C and Darrow_forwardWrite down the explicit structures of A, B, C and D in accordance with the reactions given in the synthesis plan, which shows the synthesis of D, a pheromone of moth.arrow_forward
- Give a clear handwritten answer....give the mechanism of given bleow reaction and choose the correct option...arrow_forwardCompound F may be synthesised by the method attached Draw the isomer of compound B and explain which one would be the major product and why.arrow_forwardGive the structure of compound A to D in the following reaction sequence.arrow_forward
- Give the chemical name of the products A and B in the following sequence of reactions:arrow_forwardA. Arrange the following radicals in order of decreasing rate of bromination. Justify your answer. B. Trehalose and isomaltose are both dimers of glucose. However, they have considerablydifferent reactivities. Concisely explain why these differences are observed. -Isomaltose is a reducing sugar while trehalose is not.-Trehalose is very resistant to acid hydrolysis while isomaltose can be acid-hydrolyzed withease.arrow_forwardGive the clear handwritten answer...give the mechanism for given below reactions..arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
