
Interpretation:
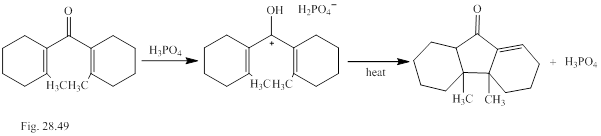
Ions as well as neutral molecules undergo pericyclic reactions. Classify the pericyclic reactions of the cation involved in the transformation shown in Fig. P28.49.The methyl groups are cis or trans is to be explained.
Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner. In phase orbital overlap results in symmetry allowed electrocyclic reactions. Selection rules of electrocyclic reactions are;
| No. of electrons | Activation mode | Stereochemistry of rotation |
| 4n | Thermal Photochemical |
Con Dis |
| 4n + 2 | Thermal Photochemical |
Dis Con |
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- Explain why compound A will not undergo a ring-opening reaction under thermal conditions, but compound B will.arrow_forwardDraw the arrow pushing mechanism for the reaction of camphor to isoborneol. For each step on the mechanism give one DETAILED sentence on what is occurring and why. Examples: "Loss of water to form a tertiary carbocation", "nucleophilic attack of ____ to increase acidity of ____", "methyl shift of ____ to form ____", etc. Thank you!arrow_forwardA. Arrange the following radicals in order of decreasing rate of bromination. Justify your answer. B. Trehalose and isomaltose are both dimers of glucose. However, they have considerablydifferent reactivities. Concisely explain why these differences are observed. -Isomaltose is a reducing sugar while trehalose is not.-Trehalose is very resistant to acid hydrolysis while isomaltose can be acid-hydrolyzed withease.arrow_forward
- Compound F may be synthesised by the method attached Draw the isomer of compound B and explain which one would be the major product and why.arrow_forwardHow does bromobenzene react differently from benzyl chloride under SN1 and SN2 conditions, and why?arrow_forwardWhich of one gives the name of the triene which when treated with an excess of perbenzoic acid gives the triepoxide shown?arrow_forward
- Compare Electrophilic aromatic substitution reactivity to quinoline and isoquinoline. Please, explain with diagramsarrow_forwardWhich of the following is statements is/are TRUE about the experiment on the relative rates of electrophilic aromatic substitution?I. The experiment must be performed in dark conditions. II. Using the same solvents, a faster decolorization will be observed in aniline as compared to ethylbenzene. III. Chlorobenzene will react faster than methoxybenzene. IV. The use of AlCl3 in the halogenation of aromatic compounds using elemental bromine could hasten the reaction.arrow_forwardWhen the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.arrow_forward
- Rationalize the selectivity observed in the benzil reduction. Draw the transition states of the nucleophilic attack leading to either meso or racemic products and indicate why one is more stable than the other.arrow_forwardAccount for the following:(i) Aniline does not give Friedel-Crafts reaction.(ii) Ethylamine is soluble in water whereas aniline is not.(iii) pKb of methylamine is less than that of aniline.arrow_forwardShow the structure of bromonium cation formed as an intermediate in bromination of trans-cinnamic acid. Clearly show stereochemistry at asymmetric carbons. Show, at which carbon nucleophilic attack of bromine anion is more likely. Explain why.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

