
Concept explainers
(a)
Interpretation:
Explain why the following equilibrium lies far to the right.
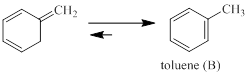
Concept introduction:
In chemistry, resonance is a systematic way to describe bonding nature of certain molecules of ions (cation or anion) by combining several structures which are contributing to the core structure. The various structures derived for a molecule is referred as resonance structures or canonical structures.
(b)
Interpretation:
Chemists had always assumed that this reaction would be so fast that compound A could never be isolated. However, this compound was prepared in 1962 and shown to be stable in the gas phase at 70°C, despite the favorable equilibrium constant for its transformation to B.Show why the conversion of A into B above would not be expected to occur as a concerted reaction.
Concept introduction:
Generally, Sigmatropic reaction is referred as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement.
(c)
Interpretation:
The concerted mechanism for the following reaction is to be explained.
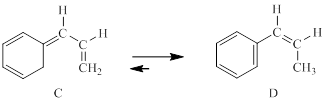
Concept introduction:
Generally, Sigmatropic reaction is referred as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. Though, the position of π-bond is changed in Sigmatropic reaction, the total number of π-bonds remain unchanged. The sigma bond can be cleaved at the middle or at the end of the π-system. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- Give the reagents and conditions necessary for the following conversion. A to B, B to D, B to C, B to E, E to F, E to G, G to H, H to I. Hence deduce the name and structural formula of the compounds C and I. Compare the procedure for converting F and E to G.arrow_forwardAlthough useful in agriculture as a soil fumigant, methyl bromide is an ozone-depleting chemical, and its production is being phased out. The industrial preparation of methyl bromide is from methanol, by reaction with hydrogen bromide. Write a mechanism for this reaction and classify it as SN1 or SN2.arrow_forwardSuggest a suitable mechanism for this multi-step reaction, and give products A and Barrow_forward
- 3c)Referring to the intermediates you drew in problem below explain in detail why no meta product is obtained in the Friedel-Crafts alkylation of chlorobenzene. Draw all pertinent resonance structures to support your argument.arrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. What is the IUPAC name for Compound X? What type of reaction (s) is/are represented by (i) and (ii)? Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B].arrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questionsA)What is the IUPAC name for Compound X?B)What type of reaction (s) is/are represented by (i) and (ii)?C)Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B]. D)Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed.E)Which of the compounds in the following pairs will occur in relatively higher yields and why?I)[1] and [2] II)[3] and [4]arrow_forward
- Below is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. –a. What is the IUPAC name for Compound X? b. What type of reaction (s) is/are represented by (i) and (ii)? c. Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B]. d. Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed.e. Which of the compounds in the following pairs will occur in relatively higher yields and why?i. [1] and [2] ii. [3] and [4]arrow_forwardBelow is a schematic representation of possible reactions that Compound X can undergo. Use the scheme to answer the following questions. A. What is the IUPAC name for Compound X? B. What type of reaction (s) is/are represented by (i) and (ii)? C. Compound X undergo transitions through either [A] or [B] to produce compounds [1], [2], [3] and [4]. Draw the structures of [A] and [B]. D. Illustrating with reaction mechanisms, show how compounds [1], [2], [3] and [4] are formed. E. Which of the compounds in the following pairs will occur in relatively higher yields and why? [1] and [2] [3] and [4] The attached image contains the scheme.arrow_forwardBelow is a schematic representation of possible reaction that compound X can undergo.Use the scheme to answer the following questions. A.What is the IUPAC name for compound X B. What type of reactions is/are represented by (i) and(ii). C.Compound X undergo transitions through either (A) or (B) to produce compounds (1),(2),(3) and (4). Draw the structure of (A) and (B).arrow_forward
- The reaction of 3,4-dimethyl-3-hexanol (3,4-dimethylhexan-3-ol) with HBr generates compound A as the major product. Treatment of compound A with a strong base gives two isomers of compound B as the major product, along with one isomer of compound C and one isomer of compound D as minor products, all of which have one double-bond equivalent. Identify compounds A, B, C, and D and give their names. By what mechanism does the reaction of 3,4-dimethyl-3-hexanol with HBr occur? By what mechanism does the reaction of A with strong base to form B occur? Propose reaction conditions for an alternative, one-step method for converting 3,4-dimethyl-3-hexanol directly to compound B.arrow_forwardGiven the sequence of reactions shown below assume the reactions are stoichiometric fill in the reagents needed for steps A B and C and explain what happens in each step of the reactions, by also namearrow_forwardWhich compound (i or ii) is the stronger base? Discuss your answer comprehensively by amoungst other providing an acid base reaction for one of the compounds.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





