
Interpretation:
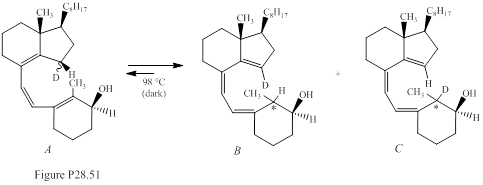
In 1985, two researchers at the University of California, Riverside, carried out the reaction given in Fig. P28.51.The equilibrium mixture contained compound A (22%), a single stereoisomer of B (47%), and a single stereoisomer of C (31%).The stereochemistry of compounds B and C at the carbon marked with the asterisk (*) is to be predicted.
Concept introduction:
Sigmatropic reaction can be described as the migration of allylic sigma bond at one end of the π-electron system to the other end of the π-electron system as an uncatalyzed intramolecular reaction. The formation of sigma bond at 3, 3-position of a 1, 5-diene is called as cope rearrangement. Notably, [3, 3] sigmatropic reaction of allyl vinyl ether is termed as Claisen rearrangement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- Compound F may be synthesised by the method attached Draw the isomer of compound B and explain which one would be the major product and why.arrow_forwardProvide TWO Grignard reactions, A and B, that can be to make the provided target structure. Give the organic reactant and the Grignard reagent that would be used, yo not need to specify the aqueous acid workup step, but you must specify which of the c-c bonds is being made in the provided traget structure. Ignore stereochemistry in this question.arrow_forwardIn the Friedel-Crafts alkylation of benzene, dialkylation is often a significant by-product. In the Friedel-Crafts acylation of benzene, diacylation is not a significant by-product. Which of the following is the primary reason for this difference? a. Acyl cations are more difficult to make with Lewis acids b.Unlike acyl cations, carbocations can undergo rearrangements. c.Alkyl groups activate the ring to further substitution, acyl groups deactivate it. d. Alkyl groups activate the ring to further substitution, acyl groups deactivate itarrow_forward
- With reference to its molecular orbital diagram, state –with justification – how CO can act as a nucleophile, and whether it will attack its reacting partner through the carbon or oxygen atomarrow_forwardShow the structure of bromonium cation formed as an intermediate in bromination of trans-cinnamic acid. Clearly show stereochemistry at asymmetric carbons. Show, at which carbon nucleophilic attack of bromine anion is more likely. Explain why.arrow_forwardGive a clear handwritten answer with explanation..give the SN2 mechanism of given bleow reactionarrow_forward
- give explanation in detailarrow_forwardOutline a synthesis of each of the following compounds from isopropyl alcohol. A compound prepared in one part can be used as a reactant in another. (Hint: which of the compounds shown can serve as a starting material to all others?)arrow_forwardWhich of the following is compatible with a Friedel-Crafts reaction?arrow_forward
- in part b. isomer of compound B. Which one would be the major product and why?arrow_forwardWhich compound (i or ii) is the stronger base? Discuss your answer comprehensively by amoungst other providing an acid base reaction for one of the compounds.arrow_forwardCompare Electrophilic aromatic substitution reactivity to quinoline and isoquinoline. Please, explain with diagramsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

