
Concept explainers
(a)
Interpretation:
The stereochemistry of compounds B and C in Fig. P28.31 is to be predicted.
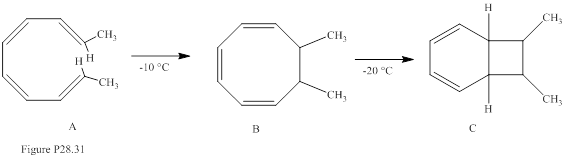
Concept introduction:
Electrocyclic reactions are a pericyclic reaction which occur intramolecularly. These reactions will result in the formation of ring compounds under the influence of heat or light. Notably, in this process one new sigma bond is formed and one old π-bond is consumed. Intriguingly, the reverse ring opening electrocyclic reaction can also be possible to occur under the same reaction mechanism but in reverse manner. Selection rules of electrocyclic reactions are;
| No. of electrons | Activation mode | Stereochemistry of rotation |
| 4n | Thermal Photochemical |
Con Dis |
| 4n + 2 | Thermal Photochemical |
Dis Con |
(b)
Interpretation:
The stereoisomer of A also gives compound C on heating is to be stated.
Concept introduction:
When the diene is having the oxygen at C3 or C4 position the sigmatropic reaction is called as oxy cope reaction. Notably, when the alkoxide fragment is involved it is referred as anionic oxy cope reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry
- Compound X was soluble in water and ether, and its aqueous solution turned litmus blue. It reacted with sodium to give a gas. The compound reacted with benzenesulfonyl chloride and base to give an insoluble product, which was unchanged with acidification. It reacted with nitrous acid to give a yellow solid. Compound A could bearrow_forwardCompound W shows stereoisomerism. Why are such isomers formed in approximately equimolar quantities no matter the synthetic pathway used for the preparation of the compound?arrow_forwardCompare the chemical structure of compound A with that of compound B. Answer it in a detailed way. Thank You.arrow_forward
- Write down the explicit structures of A, B, C and D in accordance with the reactions given in the synthesis plan, which shows the synthesis of D, a pheromone of moth.arrow_forwardIdentify the hemiacetal function in lactose and show mechanistically how the stereochemistry at that carbon can easily invert, especially in the presence of acids and bases.arrow_forwardGive the structure of the following compound(a) isobutyl bromidearrow_forward
- Naproxen is an anti-inflammatory that is the active ingredient of Aleve. Choose the compound that would be suitable for preparing the necessary diastereomeric salts to purify the racemic mixture of naproxen shown belowarrow_forwardWhen the nitrogen-containing aromatic heterocyclic compounds 1 and 2 are treated with HCl, only 1 forms the hydrochloride salt, whereas compound 2 is unreactive. Provide an explanation for this observed reactivity.arrow_forwardgive explanation in detailarrow_forward
- The sex attractant of the female housefly (Musca domestica) is called muscalure, and its structure follows. Outline a synthesis of muscalure, using the Wittig reaction. Will your synthesis lead to the required cis isomer?arrow_forwardGive a clear handwritten answer with explanation...give the all possible stereoisomers for given bleow structuresarrow_forwardBriefly outline how the enantiomers of phenylsuccinic acid will be separated from each other, starting from racemic phenylsuccinic acid to isolating (+)-phenylsuccinic acid. Explain the purpose of each step.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

