
Eight fluid ounces (1 qt = 32 oz) of a beverage in a glass at 18.0°C is to be cooled by adding ice and stirring. The properties of the beverage may be taken to be those of liquid water. The enthalpy of the ice relative to liquid water al the triple point is −348 kJ/kg. Estimate the mass of ice (g) that must melt to bring the liquid temperature to 4°C, neglecting energy losses to the surroundings. (Note: For this isobaric batch process, the energy balance reduces to 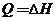
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK ELEMENTARY PRINCIPLES OF CHEMICAL P
Additional Science Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Introduction to Java Programming and Data Structures, Comprehensive Version (11th Edition)
Introduction To Programming Using Visual Basic (11th Edition)
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Java: An Introduction to Problem Solving and Programming (7th Edition)
- Define the joule in terms of SI base units.arrow_forwardIf you want to convert 56.0 g ice (at 0 °C) to water at 75.0 °C, calculate how many grams of propane, C3H8, you would have to bum to supply the energy to melt the ice and then warm it to the final temperature (at 1 bar).arrow_forwardThe enthalpy change when 1 mol methane (CH4) is burned is 890 kJ. It takes 44.0 kJ to vaporize 1 mol water. What mass of methane must be burned to provide the heat needed to vaporize 1.00 g water?arrow_forward
- What mass (g) of ethanol, CH3CH2OH(), can be vaporized at its boiling point of 78.4 C by transfer of 500. kJ to the liquid? The vapH of ethanol is 38.6 kJ/mol at this temperature.arrow_forwardAre changes in state physical or chemical changes? Explain. What type of forces must be overcome to melt or vaporize a substance (are these forces intramolecular or intermolecular)? Define the molar heat of fusion and molar heat of vaporization. Why is the molar heat of vaporization of water so much larger than its molar heat of fusion? Why does the boiling point of a liquid vary with altitude?arrow_forwardSulfur dioxide is produced in enormous amounts for sulfuric acid production. It melts at −73.0°C and boils at −10.0°C. Its ΔHfus is 8.619 kJ/mol, and its ΔHvap is 25.73 kJ/mol. The specific heat capacities of the liquid and gas are 0.995 J/g·K and 0.622 J/g·K, respectively. How much heat is required to convert 5.000 kg of solid SO2 at the melting point to a gas at 60.0 degrees C? Answer should be in J.arrow_forward
- When 4 grams of a certain compound is dissolved in 61 grams of water in a calorimeter, the temperature of the calorimeter changes from 25.00ºC to 26.98 ºC. Assuming that the specific heat capacity for the resulting solution is 4.184 J/(g oC), and the heat for this solvation is found as x kJ, what is the value of x?arrow_forwardWhat is the resulting temperature when 100 g of water at 75°C is mixed with 30 g of ice at 0 °C? Assume that all of the ice has melted. The specific heat of water is 4.184 J g-1 C°.-1 and the heat of fusion of ice is 335 J g-1.arrow_forward● One mole of water weighs 18 g at 4 °C. True Falsearrow_forward
- Ten pounds of CO2 at room temperature (80 °F) are stored in a fire extinguisher that has a volume of 4.0 ft3. How much heat must be transferred from the extinguisher so that 40 % of the CO2 becomes liquid?arrow_forwardPredict the temperature at which CuSO4·5H2O undergoes dehydration. CuSO4·5H2O (s) <=> CuSO4 (s) + 5H2O (g) ΔfHo -2280 -771 -242 kJ mol-1 Smo 300 109 189 J K-1 mol-1 T >= ______ K. 3 sig. fig.arrow_forwardIf 100.0 g H2O at 20.1 °C is mixed with 100.0 g H2O at 98.4 °C, what is the final temperature of the solution mixture ? Assume no heat is lost to the vessel in which the mixing takes place. Use CH2O = 1.00 cal/g×°Carrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning




