
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 18, Problem 30PS
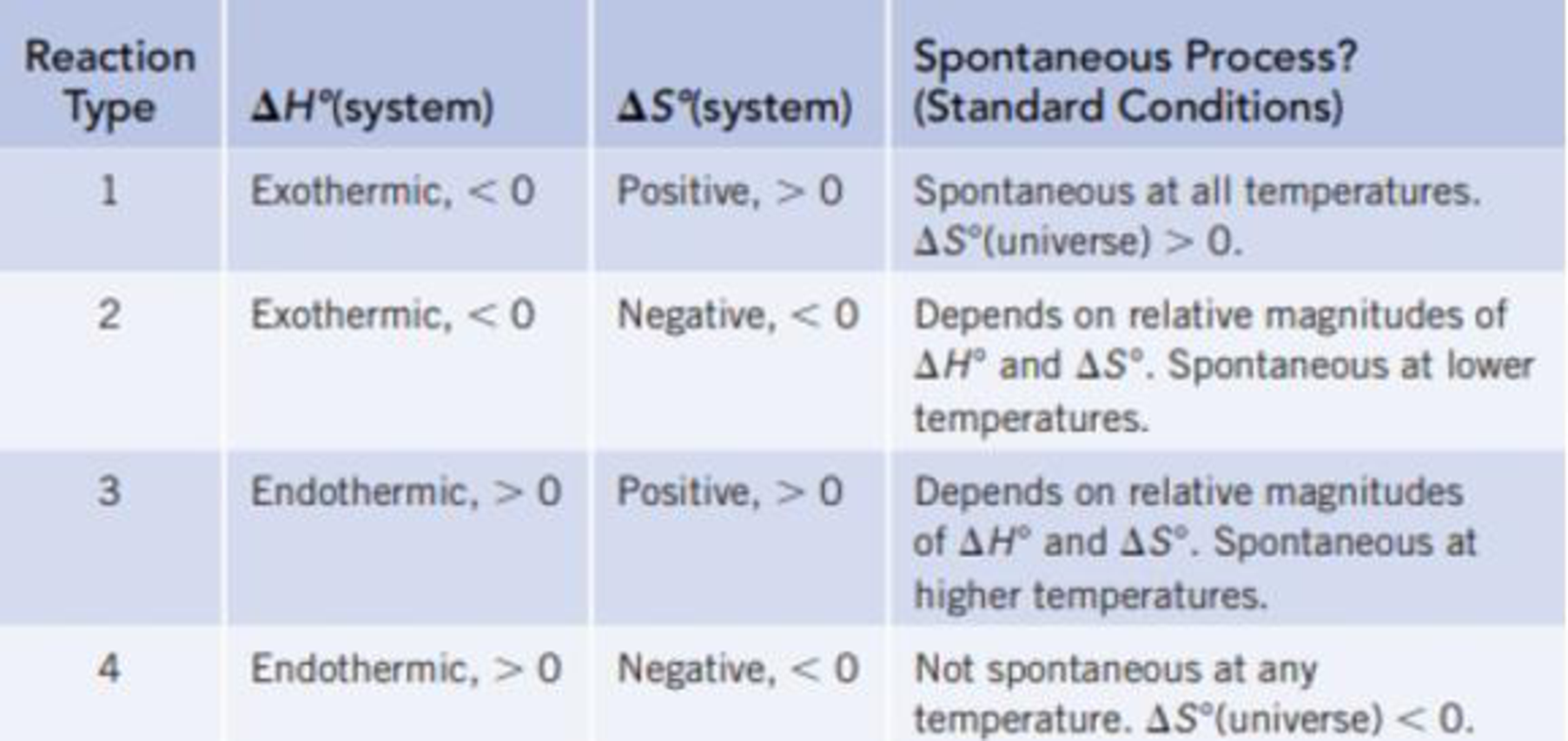
Classify each of the reactions according to one of the four reaction types summarized in Table 18.1.
- (a) C6H12O6(s) + 6 O2(g) → 6 CO2(g) + 6 H2O(ℓ)
ΔrH° = −673 kj/mol-rxn
ΔrS° = 60.4 j/K · mol-rxn
- (b) MgO(s) + C(graphite) → Mg(s) + CO(g)
ΔrH° = 490.7 kJ/mol-rxn
ΔrS° = 197.9 J/K · mol-rxn
TABLE 18.1 Predicting Whether a Reaction Will Be Spontaneous Under Standard Conditions
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 18 Solutions
Chemistry & Chemical Reactivity
Ch. 18.3 - Predict which substance in each pair has the...Ch. 18.3 - Prob. 18.2CYUCh. 18.4 - Based on rH and rS, predict the spontaneity of the...Ch. 18.6 - Prob. 18.4CYUCh. 18.6 - Prob. 18.5CYUCh. 18.6 - Oxygen was first prepared by Joseph Priestley...Ch. 18.6 - Prob. 18.7CYUCh. 18.6 - Prob. 18.8CYUCh. 18.6 - Prob. 18.9CYUCh. 18.7 - Consider the hydrolysis reactions of creatine...
Ch. 18.7 - Prob. 1.2ACPCh. 18.7 - The decomposition of diamond to graphite...Ch. 18.7 - It has been demonstrated that buckminsterfullerene...Ch. 18 - Solid NH4NO3 is placed in a beaker containing...Ch. 18 - Acetic acid, a weak acid, was added to a beaker...Ch. 18 - Identify the following processes as either...Ch. 18 - Identify the following processes as either...Ch. 18 - Prob. 5PSCh. 18 - Predict whether each of the following processes...Ch. 18 - Indicate which of the following processes are...Ch. 18 - Prob. 8PSCh. 18 - Prob. 9PSCh. 18 - Prob. 10PSCh. 18 - Prob. 11PSCh. 18 - Calculate the entropy change that occurs when 1.00...Ch. 18 - Prob. 13PSCh. 18 - Calculate the change in entropy of a system with...Ch. 18 - The third law of thermodynamics says that a...Ch. 18 - Identify trends in S values: (a) For the halogens:...Ch. 18 - Which substance has the higher entropy? (a) dry...Ch. 18 - Which substance has the higher entropy? (a) a...Ch. 18 - Use S values to calculate the standard entropy...Ch. 18 - Use S values to calculate the standard entropy...Ch. 18 - Calculate the standard entropy change for the...Ch. 18 - Calculate the standard entropy change for the...Ch. 18 - Calculate the standard entropy change for the...Ch. 18 - Calculate the standard entropy change for the...Ch. 18 - Is the reaction Si(s) + 2 Cl2(g) SiCl4(g)...Ch. 18 - Is the reaction Si(s) + 2 H2(g) SiH4(g)...Ch. 18 - Calculate S(universe) for the decomposition of 1...Ch. 18 - Calculate S(universe) for the formation of 1 mol...Ch. 18 - Classify each of the reactions according to one of...Ch. 18 - Classify each of the reactions according to one of...Ch. 18 - Using values of fH and S, calculate rG for each of...Ch. 18 - Using values of fH and S, calculate rG for each of...Ch. 18 - Using values of fH and S, calculate the standard...Ch. 18 - Using values of fH and S, calculate the standard...Ch. 18 - Using values of fG, calculate rG for each of the...Ch. 18 - Using values of fG, calculate rG for each of the...Ch. 18 - For the reaction BaCO3(s) BaO(s) + CO2(g), rG =...Ch. 18 - For the reaction TiCl2(s) + Cl2(g) TiCl4(), rG =...Ch. 18 - Determine whether the reactions listed below are...Ch. 18 - Determine whether the reactions listed below are...Ch. 18 - Heating some metal carbonates, among them...Ch. 18 - Calculate rH and rS for the reaction of tin(IV)...Ch. 18 - The ionization constant, Ka, for acetic acid is...Ch. 18 - Prob. 44PSCh. 18 - The standard free energy change, rG, for the...Ch. 18 - Prob. 46PSCh. 18 - Calculate rG at 25 C for the formation of 1.00 mol...Ch. 18 - Prob. 48PSCh. 18 - Prob. 49PSCh. 18 - Prob. 50PSCh. 18 - Compare the compounds in each set below and decide...Ch. 18 - Using standard entropy values, calculate rS for...Ch. 18 - About 5 billion kilograms of benzene, C6H6, are...Ch. 18 - Hydrogenation, the addition of hydrogen to an...Ch. 18 - Is the combustion of ethane, C2H6, product-favored...Ch. 18 - Prob. 56GQCh. 18 - When vapors from hydrochloric acid and aqueous...Ch. 18 - Calculate S(system), S(surroundings), and...Ch. 18 - Methanol is now widely used as a fuel in race...Ch. 18 - The enthalpy of vaporization of liquid diethyl...Ch. 18 - Calculate the entropy change, rS, for the...Ch. 18 - Using thermodynamic data, estimate the normal...Ch. 18 - Prob. 63GQCh. 18 - When calcium carbonate is heated strongly, CO2 gas...Ch. 18 - Sodium reacts violently with water according to...Ch. 18 - Yeast can produce ethanol by the fermentation of...Ch. 18 - Elemental boron, in the form of thin fibers, can...Ch. 18 - Prob. 68GQCh. 18 - Prob. 69GQCh. 18 - Estimate the boiling point of water in Denver,...Ch. 18 - The equilibrium constant for the butane ...Ch. 18 - A crucial reaction for the production of synthetic...Ch. 18 - Calculate rG for the decomposition of sulfur...Ch. 18 - Prob. 74GQCh. 18 - A cave in Mexico was recently discovered to have...Ch. 18 - Wet limestone is used to scrub SO2 gas from the...Ch. 18 - Sulfur undergoes a phase transition between 80 and...Ch. 18 - Calculate the entropy change for dissolving HCl...Ch. 18 - Some metal oxides can be decomposed to the metal...Ch. 18 - Prob. 80ILCh. 18 - Prob. 81ILCh. 18 - Prob. 82ILCh. 18 - Titanium(IV) oxide is converted to titanium...Ch. 18 - Cisplatin [cis-diamminedichloroplatinum(II)] is a...Ch. 18 - Prob. 85ILCh. 18 - Explain why each of the following statements is...Ch. 18 - Decide whether each of the following statements is...Ch. 18 - Under what conditions is the entropy of a pure...Ch. 18 - Prob. 89SCQCh. 18 - Consider the formation of NO(g) from its elements....Ch. 18 - Prob. 91SCQCh. 18 - The normal melting point of benzene, C6H6, is 5.5...Ch. 18 - Prob. 93SCQCh. 18 - For each of the following processes, predict the...Ch. 18 - Heater Meals are food packages that contain their...Ch. 18 - Prob. 96SCQCh. 18 - Prob. 97SCQCh. 18 - Prob. 98SCQCh. 18 - Iodine, I2, dissolves readily in carbon...Ch. 18 - Prob. 100SCQCh. 18 - Prob. 101SCQCh. 18 - Prob. 102SCQCh. 18 - Prob. 103SCQCh. 18 - Prob. 104SCQCh. 18 - The Haber-Bosch process for the production of...Ch. 18 - Prob. 106SCQCh. 18 - Prob. 107SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Silver carbonate, Ag2CO3, is a light yellow compound that decomposes when heated to give silver oxide and carbon dioxide: Ag2CO3(s)Ag2O(s)+CO2(g) A researcher measured the partial pressure of carbon dioxide over a sample of silver carbonate at 220C and found that it was 1.37 atm. Calculate the partial pressure of carbon dioxide at 25C. The standard enthalpies of formation of silver carbonate and silver oxide at 25C are 505.9 kJ/mol and 31.05 kJ/mol, respectively. Make any reasonable assumptions in your calculations. State the assumptions that you make, and note why you think they are reasonable.arrow_forwardAdenosine triphosphate, ATP, is used as a free-energy source by biological cells. (See the essay on page 624.) ATP hydrolyzes in the presence of enzymes to give ADP: ATP(aq)+H2O(l)ADP(aq)+H2PO4(aq);G=30.5kJ/molat25C Consider a hypothetical biochemical reaction of molecule A to give molecule B: A(aq)B(aq);G=+15.0kJ/molat25C Calculate the ratio [B]/[A] at 25C at equilibrium. Now consider this reaction coupled to the reaction for the hydrolysis of ATP: A(aq)+ATP(aq)+H2O(l)B(aq)+ADP(aq)+H2PO4(aq) If a cell maintains a high ratio of ATP to ADP and H2PO4 by continuously making ATP, the conversion of A to B can be made highly spontaneous. A characteristic value of this ratio is [ATP][ADP][H2PO4]=500 Calculate the ratio [B][A] in this case and compare it with the uncoupled reaction. Compared with the uncoupled reaction, how much larger is this ratio when coupled to the hydrolysis of ATP?arrow_forwardWhat is the difference between S, S , and S 298 for a Chemical change?arrow_forward
- For the decomposition of formic acid, HCOOH(l)H2O(l)+CO(g) H = +29 kJ/mol at 25C. a Does the tendency of this reaction to proceed to a state of minimum energy favor the formation of water and carbon monoxide or formic acid? Explain. b Does the tendency of this reaction to proceed to a state of maximum entropy favor the formation of products or reactants? Explainarrow_forwardNatural gas, which is mostly methane, CH4, is a resource that the United States has in abundance. In principle, ethane can be obtained from methane by the reaction 2CH4(g)C2H6(g)+H2(g) (a) Calculate G° at 25°C for the reaction. Comment on the feasibility of this reaction at 25°C. (b) Couple the reaction above with the formation of steam from the elements: H2(g)+12O2(g)H2O(g)G=228.6kJ What is the equation for the overall reaction? Comment on the feasibility of the overall reaction.arrow_forwardAccording to a source, lithium peroxide (Li2O2) decomposes to lithium oxide (Li2O) and oxygen gas at about 195C. If the standard enthalpy change for this decomposition is 33.9 kJ/mol, what would you give as an estimate for the standard entropy change for this reaction? Explain.arrow_forward
- The free energy of formation of one mole of compound refers to a particular chemical equation. For each of the following, write that equation. a KBr(s) b CH3Cl(l) c H2S(g) d AsH3(g)arrow_forwardElemental boron, in the form of thin fibers, can be made by reducing a boron halide with H2. BCl3(g) + 32 H2(g) B(s) + 3 HCl(g) Calculate rH, rS, and rG at 25 C for this reaction. Is the reaction predicted to be product-favored at equilibrium at 25 C? If so, is it enthalpy- or entropy-driven? [S for B(s) is 5.86 J/K mol.]arrow_forwarda Calculate K1, at 25C for sulfurous acid: H2SO3(aq)H+(aq)+HSO3(aq) b Which thermodynamic factor is the most significant in accounting for the fact that sulfurous acid is a weak acid? Why?arrow_forward
- What is the change in entropy, S, for the reaction CaCO3(s)+2H(aq)Ca2+(aq)+H2O(l)+CO2(g) See Table 18.1 for values of standard entropies. Does the entropy of the chemical system increase or decrease as you expect? Explain.arrow_forwardThe combustion of acetylene, C2H2, is a spontaneous reaction given by the equation 2C2H2(g)+5O2(g)4CO2(g)+2H2O(l) As expected for a combustion, the reaction is exothermic. What is the sign of H? What do you expect for the sign of S? Explain the spontaneity of the reaction in terms of the enthalpy and entropy changes.arrow_forwardElemental boron, in the form of thin fibers, can be made by reducing a boron halide with H2. BCl3(g) + 3/2 H2(g) B(s) + 3HCl(g) Calculate H, S, and G at 25 C for this reaction. Is the reaction predicted to be product favored at equilibrium at 25 C? If so, is it enthalpy driven or entropy driven?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY