
Concept explainers
Interpretation:
The Lewis structure of the molecule whose Newman projection is given is to be drawn.
Concept introduction:
In a Newman projection, the bond of interest connecting the atoms is not shown. But the atoms connected are shown explicitly. The front atom is shown as a point while the back atom is shown as a circle. The atoms attached to the front carbon converge at the point while the atoms attached to the back carbon are shown connected to the circle.
Answer to Problem 4.1P
The Lewis structure of the molecule is:
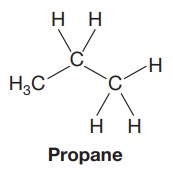
Explanation of Solution
The given Newman projection is:
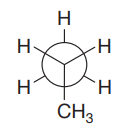
In this Newman projection, the front carbon atom is shown by a point. It is attached to two hydrogen atoms and a methyl group. The back carbon atom is shown by a circle. It is attached to three hydrogen atoms. Thus, in the Lewis structure, there will be a chain of three carbon atoms. The molecule is propane. Its Lewis structure is shown below:

The Lewis structure of the molecule is drawn as shown above.
Want to see more full solutions like this?
Chapter 4 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- Problem. Calculate the dipole moment of Glycine molecule using partial charge and positions of atoms. In this problem, How can I get the partial charge of the atoms? Should I find the reference or appendix?arrow_forwardChoose the correct Newman projection for the molecule show as viewed along the arrow.arrow_forwardFor problem 3, why does N keep its electrons in addition to taking the H+ from the other molecule? Didn't it give its electrons to the other molecule?arrow_forward
- Draw a more stable resonance structure for the following molecule. Use a curved arrow to show how to transform the original structure to the new one and please specify charges.arrow_forwardplease answer this spec question! answer choices are given except toluene and tetrahydrofuran!arrow_forwardmatch to the corresponding moleculearrow_forward
- please drW this molecule confused about the Z confgurationarrow_forwardmatch the following to its compound name. *First row only from upper to bottomarrow_forwardDraw the resonance structure for the following molecule: Just started with resonance, where would the arrows go? I'm unsure of this problem.arrow_forward
- Fill in the Newman projection for the following molecule from the angle shownarrow_forwardShow the other two resonance structures of the following molecule (show the curved arrows). Indicate the major contributor. Draw the resonance hybrid.arrow_forwardPut the lone pairs on the following structures where appropriate and draw the curved arrows on the left-hand structures that will result in the right-hand structures.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
