
Concept explainers
Interpretation:
For the given Newman projection of cyclohexane, all the
Concept introduction:
Newman projection is a two-dimensional representation of a molecule viewed down the bond of interest. In a Newman projection, two atoms directly connected by the bond of interest are shown explicitly. The nearer atom is depicted as a point while the more distant atom is depicted as a circle. Bonds of the front (nearer) atom converge at the point whereas bonds to the back atom connect to the circle. The bond of interest is not visible; instead it must be imagined as connecting the front and the back carbon atoms. In cyclohexane, all six carbon atoms are completely indistinguishable, but there are two different types of hydrogen atoms; six hydrogen atoms occupy the equatorial positions and six occupy the axial position. Each carbon atom in cyclohexane is bonded to one of each. The basic framework of chair form of cyclohexane is:
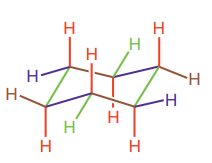
Axial bonds are alternate up and worn around the ring (highlighted in red). Equatorial bonds are alternate slightly up and down around the ring (highlighted in blue).
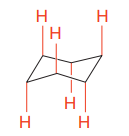
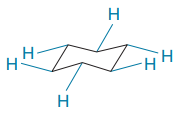
In the rotational conformations, the bonds which are up remain up and the bonds which are down remain down.
Trending nowThis is a popular solution!

Chapter 4 Solutions
ORGANIC CHEMISTRY E-BOOK W/SMARTWORK5
- A trisubstituted cyclohexane with three substituents-red, green, and blue-undergoes a ring-flip to its alternate chair conformation. Identify each substituent as axial or equatorial, and show the positions occupied by the three substituents in the ring-flipped form.arrow_forwardFollowing is a chair conformation of cyclohexane with the carbon atoms numbered 1 through 6. (a) Draw hydrogen atoms that are above the plane of the ring on carbons 1 and 2 and below the plane of the ring on carbon 4. (b) Which of these hydrogens are equatorial? Which are axial? (c) Draw the alternative chair conformation. Which hydrogens are equatorial? Which are axial? Which are above the plane of the ring? Which are below it?arrow_forwardConsider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.arrow_forward
- Draw (a) a Newman projection of the most stable conformation sighting down the C-3 C-4 bond and (b) a bond-line depiction of 2,2,5,5-tetramethylhexane.arrow_forwardDraw the Newman projections of 3-methylpentane looking down C2-C3 bond. Identify the most stable conformationarrow_forwardWhich Newman projection is the most stable conformer for (S)-4-chlorobutan-2-ol?arrow_forward
- Draw the Newman projection of the MOST stable and LEAST stable conformation. along in the C2-C3 bond. 3- Methyloctane 2,2-Dimethylbutane 2,3,4-Trimethylhexanearrow_forwardSighting along the C2-C3 bond of the 2-methylbutane, draw the Newman projection ofconformational isomer. State the most stable conformerarrow_forwardDraw the Newman projection of the boat conformation of Methylcyclohexane.arrow_forward
- Use Newman projection to draw 1-chloro-2-methylbutane in its most stable conformation, looking down from C-2 to C-3. Hint: draw the line angle formula first.arrow_forwardDraw the Newman projection of the methylcyclohexane looking along the C1 – C2 and C5 – C4 bonds.arrow_forwardSight along the C2-C3 bond of 2, 3-dimethylbutane, and draw a Newman projection of the most stable conformation.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


